Essential idea: The continued survival of living organisms including humans depends on sustainable communities.
4.1 Species, communities and ecosystems
UNDERSTANDINGS:
U4.1.1 Species are groups of organisms that can potentially interbreed to produce fertile offspring.
Species: a group of organisms that can interbreed and produce fertile offspring. Spec point is self explanatory.
U4.1.2 Members of a species may be reproductively isolated in separate populations.
It is commonly assumed that members of one species are reproductively isolated from members of all other species (i.e. they cannot mate with them to produce fertile offspring). However, we must be cautious in defining species with this criterion because members of very closely related species can sometimes produce offspring together and a small fraction of those may be fertile to some degree. This is the case with mules, which are the product of mating between horses and donkeys. About one out of 10,000 mules is fertile. This suggests that some species differences are a matter of degree.
U4.1.3 Species have either an autotrophic or heterotrophic method of nutrition (a few species have both methods).
Organisms are divided into autotrophs and heterotrophs according to their energy pathways. Autotrophs are those organisms that are able to make energy-containing organic molecules from inorganic raw material by using basic energy sources such as sunlight. Plants are the prime example of autotrophs, using photosynthesis.
So, autotrophs are organisms that synthesise their organic molecules from simple inorganic substances (can produce their own food) in their surroundings using light (photosynthesis) or chemical energy (chemosynthesis).
All other organisms must make use of food that comes from other organisms in the form of fats, carbohydrates and proteins. These organisms which feed on others are called heterotrophs. They are organisms that obtain organic molecules from other organisms.
UNDERSTANDINGS:
U4.1.1 Species are groups of organisms that can potentially interbreed to produce fertile offspring.
Species: a group of organisms that can interbreed and produce fertile offspring. Spec point is self explanatory.
U4.1.2 Members of a species may be reproductively isolated in separate populations.
It is commonly assumed that members of one species are reproductively isolated from members of all other species (i.e. they cannot mate with them to produce fertile offspring). However, we must be cautious in defining species with this criterion because members of very closely related species can sometimes produce offspring together and a small fraction of those may be fertile to some degree. This is the case with mules, which are the product of mating between horses and donkeys. About one out of 10,000 mules is fertile. This suggests that some species differences are a matter of degree.
U4.1.3 Species have either an autotrophic or heterotrophic method of nutrition (a few species have both methods).
Organisms are divided into autotrophs and heterotrophs according to their energy pathways. Autotrophs are those organisms that are able to make energy-containing organic molecules from inorganic raw material by using basic energy sources such as sunlight. Plants are the prime example of autotrophs, using photosynthesis.
So, autotrophs are organisms that synthesise their organic molecules from simple inorganic substances (can produce their own food) in their surroundings using light (photosynthesis) or chemical energy (chemosynthesis).
All other organisms must make use of food that comes from other organisms in the form of fats, carbohydrates and proteins. These organisms which feed on others are called heterotrophs. They are organisms that obtain organic molecules from other organisms.
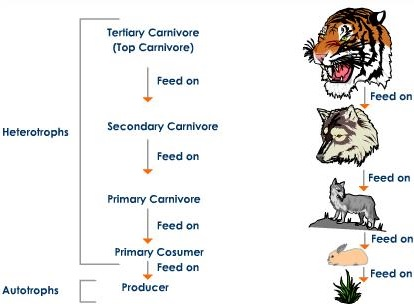
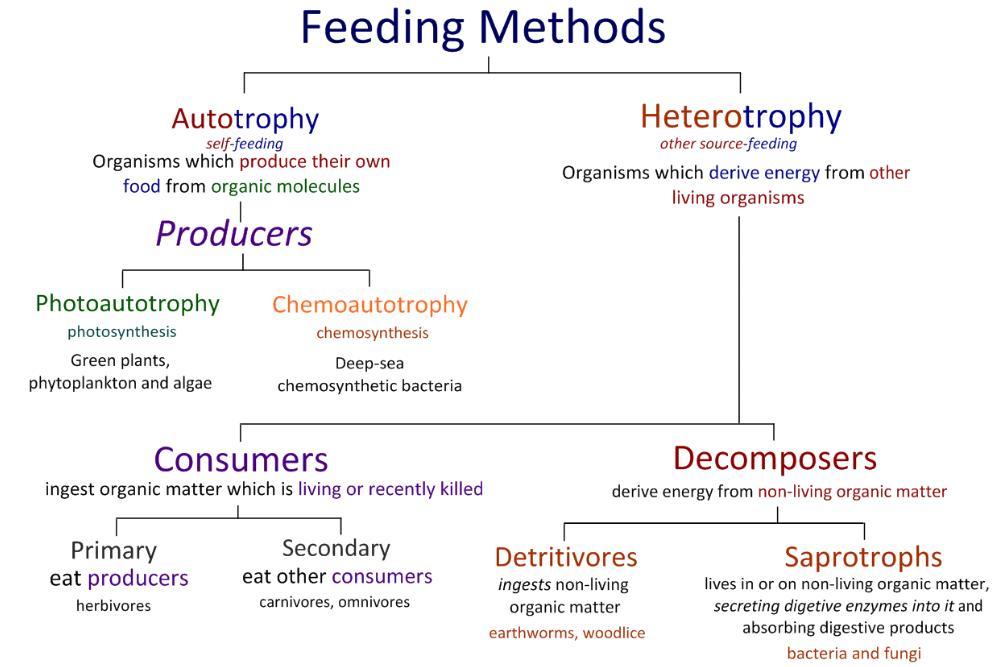
U4.1.4 Consumers are heterotrophs that feed on living organisms by ingestion.
Living things require energy to drive the activities of life, but energy transfer through organisms is one-way. Species have either autotrophic or heterotrophic methods of nutrition. Consumers (an organism that eats another organism) are heterotrophs that feed on living organisms by ingestion. In food chains, energy is passed from producers (photosynthetic green plants) to primary consumers (herbivores), to secondary and tertiary consumers (carnivores).
Consumers eat organisms that are either alive or organisms that have been dead for a short period of time.
U4.1.5 Detritivores are heterotrophs that obtain organic nutrients from detritus by internal digestion.
Organisms produce waste in the form of large quantities of organic matter, such as dead leaves (and other parts of plants); feathers, hairs and dead parts of an animal's body as well as and faeces from animals. If this were to accumulate in an ecosystem, it would become toxic, so instead, it is used as a source of nutrition by two groups of heterotroph organisms: detritivores and saprotrophs.
Detritivores are heterotrophs that obtain organic nutrients from detritus by internal digestion. These organisms ingest dead organic matter, digest it internally and the products of digestion assimilate into their bodies. This can be done in different ways. Larger, multicellular detritivores such as earthworms, absorb the dead organic matter into their gut, while unicellular organisms ingest it into food vacuoles, where, as always it is broken down by enzymes.
U4.1.6 Saprotrophs are heterotrophs that obtain organic nutrients from dead organisms by external digestion.
Differing from detritivores, saprotrophs are heterotrophs that obtain organic nutrients from dead organic matter by external digestion. They do this by secreting digestive enzymes into the dead organic matter and thus digesting it externally and absorbing the products of such digestion. Many types of bacteria and fungi are saprotrophic, also known as decomposers because of the breaking down of carbon compounds in dead organic matter and release of elements such as nitrogen into the ecosystem-which are then used again by other organisms.
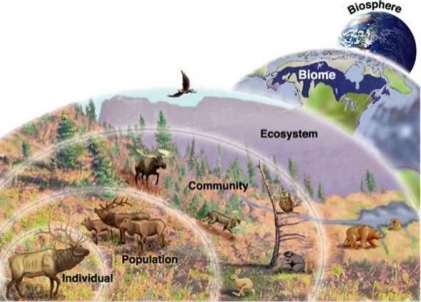
U4.1.7 A community is formed by populations of different species living together and interacting with each other.
- Communities are formed by populations of different species living together and interacting with each other
- There are many types of relationships between different organisms
- These include competition: when two species fight for a common need (such as food or habitat)
a) MUTUALISM (+ , +)
* a symbiotic relationship in which both organisms BENEFIT
ex. nitrogen fixing bacteria that live in nodes (lumps) on the roots of certain plants (legumes)...the bacteria have a nice place to live (+), and the plants benefit from getting the nitrogen they need from the bacteria (+)
b) COMMENSALISM (+ , 0)
* a symbiotic relationship where one organism benefits (+) and the otherorganism is not harmed (0)
ex. the remora (a small fish) attaches itself to the underside of a shark...when the shark feeds, the remora disconnects and eats scraps that are left over (+) ... the shark is not affected (0); barnacles (+) on whales (0)
c) PARASITISM (+ , -)
* a symbiotic relationship where one organism, the parasite, benefits (+), while the other organism, the host, is harmed (-)
ex. athlete's foot, a fungus, grows on human feet for nutrients (+), while the human doesn't like it (-);
tapeworms (+) in humans (-); heartworms (+) in dogs (-)
d) PREDATOR-PREY (+, -)
* a symbiotic relationship where one organism eats another!
ex. predator--lions (+), prey--gazelles (-)
- Either good or bad relationship, a specie cannot thrive without interactions and live in isolation.
U4.1.8 A community forms an ecosystem by its interactions with the abiotic environment.
Ecosystems are made up of the interactions between the living and non-living components within them. It is impossible to think of an ecosystem without including these interactions. The living components of an ecosystem are known as the “biotic factors” - living biological factors that influence the other organsims or environment of an ecosystem.
This is a lot more than just listing the plants, animals or micro-organisms found in an ecosystem. It includes the roles played by the organisms. Biotic factors interact as:
- producers
- consumers
- detrivores
- decomposers
- parasite
- host
- predator
- competitor
- herbivore
- carnivore
- symbiant
- pathogen
A tree in a woodland is a producer providing the basic unit of energy for the rest of the ecosystem. But at the same time it competes for light with other trees and may be the host to parasitic plants such as mistletoe or decomposing fungi. During the annual cycle in the wood, the tree will at times take water and mineral nutrients from the soil and at others return nutrients from fallen wood and leaves.
Abotic factors:
- are the non-living parts of the environment
- can vary from place to place
- directly affect the ability of organisms to live and reproduce
Abiotic factors can act as limiting factors that keep a population at a certain level
ex. desert environment -- hot temperature and little water are examples of limiting factors -- different species living in the desert are limited mainly to those types of plants and animals that need very little water and can survive extreme temperatures.
Abiotic factors include:
- the atmosphere
- climate and water
- soil structure and chemistry
- water chemistry
- seasonality
U4.1.9 Autotrophs obtain inorganic nutrients from the abiotic environment.
U4.1.10 The supply of inorganic nutrients is maintained by nutrient cycling.
Chemical elements are able to be recycled continuously, which explains why living organisms do not run out of them. Organisms absorb the elements they need as inorganic nutrients from the abiotic environment, use them and then return them, without altering the atoms or their chemical structure.
Living things require energy to drive the activities of life, but energy transfer through organisms is one-way. Species have either autotrophic or heterotrophic methods of nutrition. Consumers (an organism that eats another organism) are heterotrophs that feed on living organisms by ingestion. In food chains, energy is passed from producers (photosynthetic green plants) to primary consumers (herbivores), to secondary and tertiary consumers (carnivores).
Consumers eat organisms that are either alive or organisms that have been dead for a short period of time.
U4.1.5 Detritivores are heterotrophs that obtain organic nutrients from detritus by internal digestion.
Organisms produce waste in the form of large quantities of organic matter, such as dead leaves (and other parts of plants); feathers, hairs and dead parts of an animal's body as well as and faeces from animals. If this were to accumulate in an ecosystem, it would become toxic, so instead, it is used as a source of nutrition by two groups of heterotroph organisms: detritivores and saprotrophs.
Detritivores are heterotrophs that obtain organic nutrients from detritus by internal digestion. These organisms ingest dead organic matter, digest it internally and the products of digestion assimilate into their bodies. This can be done in different ways. Larger, multicellular detritivores such as earthworms, absorb the dead organic matter into their gut, while unicellular organisms ingest it into food vacuoles, where, as always it is broken down by enzymes.
U4.1.6 Saprotrophs are heterotrophs that obtain organic nutrients from dead organisms by external digestion.
Differing from detritivores, saprotrophs are heterotrophs that obtain organic nutrients from dead organic matter by external digestion. They do this by secreting digestive enzymes into the dead organic matter and thus digesting it externally and absorbing the products of such digestion. Many types of bacteria and fungi are saprotrophic, also known as decomposers because of the breaking down of carbon compounds in dead organic matter and release of elements such as nitrogen into the ecosystem-which are then used again by other organisms.
U4.1.7 A community is formed by populations of different species living together and interacting with each other.
- Communities are formed by populations of different species living together and interacting with each other
- There are many types of relationships between different organisms
- These include competition: when two species fight for a common need (such as food or habitat)
a) MUTUALISM (+ , +)
* a symbiotic relationship in which both organisms BENEFIT
ex. nitrogen fixing bacteria that live in nodes (lumps) on the roots of certain plants (legumes)...the bacteria have a nice place to live (+), and the plants benefit from getting the nitrogen they need from the bacteria (+)
b) COMMENSALISM (+ , 0)
* a symbiotic relationship where one organism benefits (+) and the otherorganism is not harmed (0)
ex. the remora (a small fish) attaches itself to the underside of a shark...when the shark feeds, the remora disconnects and eats scraps that are left over (+) ... the shark is not affected (0); barnacles (+) on whales (0)
c) PARASITISM (+ , -)
* a symbiotic relationship where one organism, the parasite, benefits (+), while the other organism, the host, is harmed (-)
ex. athlete's foot, a fungus, grows on human feet for nutrients (+), while the human doesn't like it (-);
tapeworms (+) in humans (-); heartworms (+) in dogs (-)
d) PREDATOR-PREY (+, -)
* a symbiotic relationship where one organism eats another!
ex. predator--lions (+), prey--gazelles (-)
- Either good or bad relationship, a specie cannot thrive without interactions and live in isolation.
U4.1.8 A community forms an ecosystem by its interactions with the abiotic environment.
Ecosystems are made up of the interactions between the living and non-living components within them. It is impossible to think of an ecosystem without including these interactions. The living components of an ecosystem are known as the “biotic factors” - living biological factors that influence the other organsims or environment of an ecosystem.
This is a lot more than just listing the plants, animals or micro-organisms found in an ecosystem. It includes the roles played by the organisms. Biotic factors interact as:
- producers
- consumers
- detrivores
- decomposers
- parasite
- host
- predator
- competitor
- herbivore
- carnivore
- symbiant
- pathogen
A tree in a woodland is a producer providing the basic unit of energy for the rest of the ecosystem. But at the same time it competes for light with other trees and may be the host to parasitic plants such as mistletoe or decomposing fungi. During the annual cycle in the wood, the tree will at times take water and mineral nutrients from the soil and at others return nutrients from fallen wood and leaves.
Abotic factors:
- are the non-living parts of the environment
- can vary from place to place
- directly affect the ability of organisms to live and reproduce
Abiotic factors can act as limiting factors that keep a population at a certain level
ex. desert environment -- hot temperature and little water are examples of limiting factors -- different species living in the desert are limited mainly to those types of plants and animals that need very little water and can survive extreme temperatures.
Abiotic factors include:
- the atmosphere
- climate and water
- soil structure and chemistry
- water chemistry
- seasonality
U4.1.9 Autotrophs obtain inorganic nutrients from the abiotic environment.
U4.1.10 The supply of inorganic nutrients is maintained by nutrient cycling.
Chemical elements are able to be recycled continuously, which explains why living organisms do not run out of them. Organisms absorb the elements they need as inorganic nutrients from the abiotic environment, use them and then return them, without altering the atoms or their chemical structure.
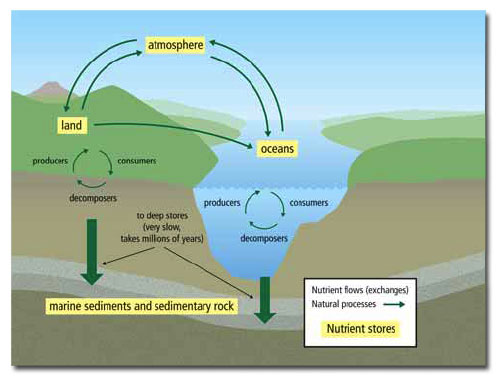

U4.1.11 Ecosystems have the potential to be sustainable over long periods of time.
APPLICATION:
N/A
SKILL:
S4.1.1 Classifying species as autotrophs, consumers, detritivores or saprotrophs from a knowledge of their mode of nutrition.
See U4.1.4, U4.1.5 and U4.1.6.
S4.1.2 Setting up sealed mesocosms to try to establish sustainability. (Practical 5)
A mesocosm is an experimental tool that brings a small part of the natural environment under controlled conditions. In this way mesocosms provide a link between observational field studies that take place in natural environments, but without replication, and controlled laboratory experiments that may take place under somewhat unnatural conditions.
When pursuing a laboratory experiment, the experimenter cannot account for every possible factor that would normally occur in the original environment. Mesocosms circumvent this problem as the experiment is performed in the natural environment, but in an enclosure that is small enough that key variables can be brought under control. Mesocosms have been used to evaluate how organisms or communities might react to environmental change, through deliberate manipulation of environmental variables, such as increased temperature, carbon dioxide or pH levels.
Advantages:
The advantage to mesocosm studies provides us with what we already know and what possible influential factors will produce a positive or negative reaction of our subject areas of interest. Manipulating something can give us an idea as to what to expect if something were to occur in that ecosystem or environment. For indoor mesocosms, growth chambers allow us to control the experiment. You can place plants in a growth chamber and manipulate the air, temperature, heat and light distribution and observe the effects when exposed to different amounts of each factor.
Greenhouses also contribute to mesocosm studies although sometimes, it may induce climate change, interfering with the experiment and resulting in inefficient data.
Disadvantages:
Using growth chambers for a laboratory experiment is sometimes a disadvantage due to the limited amount of space. Another disadvantage to using mesocosms is not adequately imitating the environment, causing the organism to avoid giving off a certain reaction versus its natural behaviour in its original environment.
APPLICATION:
N/A
SKILL:
S4.1.1 Classifying species as autotrophs, consumers, detritivores or saprotrophs from a knowledge of their mode of nutrition.
See U4.1.4, U4.1.5 and U4.1.6.
S4.1.2 Setting up sealed mesocosms to try to establish sustainability. (Practical 5)
A mesocosm is an experimental tool that brings a small part of the natural environment under controlled conditions. In this way mesocosms provide a link between observational field studies that take place in natural environments, but without replication, and controlled laboratory experiments that may take place under somewhat unnatural conditions.
When pursuing a laboratory experiment, the experimenter cannot account for every possible factor that would normally occur in the original environment. Mesocosms circumvent this problem as the experiment is performed in the natural environment, but in an enclosure that is small enough that key variables can be brought under control. Mesocosms have been used to evaluate how organisms or communities might react to environmental change, through deliberate manipulation of environmental variables, such as increased temperature, carbon dioxide or pH levels.
Advantages:
The advantage to mesocosm studies provides us with what we already know and what possible influential factors will produce a positive or negative reaction of our subject areas of interest. Manipulating something can give us an idea as to what to expect if something were to occur in that ecosystem or environment. For indoor mesocosms, growth chambers allow us to control the experiment. You can place plants in a growth chamber and manipulate the air, temperature, heat and light distribution and observe the effects when exposed to different amounts of each factor.
Greenhouses also contribute to mesocosm studies although sometimes, it may induce climate change, interfering with the experiment and resulting in inefficient data.
Disadvantages:
Using growth chambers for a laboratory experiment is sometimes a disadvantage due to the limited amount of space. Another disadvantage to using mesocosms is not adequately imitating the environment, causing the organism to avoid giving off a certain reaction versus its natural behaviour in its original environment.
S4.1.3 Testing for association between two species using the chi-squared test with data obtained by quadrat sampling.
S4.1.4 Recognising and interpreting statistical significance.
S4.1.4 Recognising and interpreting statistical significance.
Essential idea: Ecosystems require a continuous supply of energy to fuel life processes and to replace energy lost as heat.
4.2 Energy Flow
UNDERSTANDINGS:
U4.2.1 Most ecosystems rely on a supply of energy from sunlight.
The most important organisms in any food chain are the producers because without them, the next trophic level would have nothing to consume. Since photosynthetic organisms occupy the first trophic level, sunlight is the initial energy source for almost all communities.
The vast majority of the foods we eat,the energy they contain can be traced back to sunlight. Milk, for example, gets its energy from the cow which produced it. The cow obtained its energy from grass, which got its energy from sunlight. In marine ecosystems, most food chains start with phytoplankton which get their energy from sunlight.
Leaves are a food source when they are green and photosynthesising and continue to be a source of food when they have fallen to the ground and turned brown. Decomposers and detritus feeders such as soil organisms or some of the bottom feeders in a river depend on this dead organic matter for food.
Sunlight is almost always the first type of energy to enter an ecosystem. Some living things, like plants, are able to capture the energy from sunlight. When another living thing in an ecosystem eats a plant, it is gaining energy that came first from the sun.
U4.2.2 Light energy is converted to chemical energy in carbon compounds by photosynthesis.
Photosynthesis is a process used by plants and other organisms to convert light energy, normally from the sun, into chemical energy that can be later released to fuel the organisms' activities. This chemical energy is stored in carbohydrate molecules, such as sugars, which are synthesised from carbon dioxide and water.
Producers absorb light energy through photosynthetic pigments which convert it to chemical energy. This chemical energy is used to create carbon compounds such as carbohydrates and lipids. These producers release energy from their carbon compounds by cell respiration, the rest remains in their cells and tissues to provide it to the heterotrophs
U4.2.3 Chemical energy in carbon compounds flows through food chains by means of feeding.
Producers are the first organisms in a food chain since they obtain energy from sunlight instead of from other organisms. Organisms that feed on producers and obtain energy from them are primary consumers in the food chain. Organisms that feed on primary consumers are secondary consumers. Organisms that feed on secondary consumers are tertiary consumers (and so on). The arrows on a food chain show the transportation of energy, which is why an arrow flows from the first organisms (producers/primary consumers) to the last (secondary/tertiary consumers).
U4.2.4 Energy released from carbon compounds by respiration is used in living organisms and converted to heat.
Producers can release energy from their carbon compounds by cell respiration and then use it for cell activities. Energy released in this way is eventually lost to the environment as waste heat. However, only some of the carbon compounds in producers are used in this way and the largest part remains in the cells and tissues of producers. The energy in these carbon compounds is available to heterotrophs.
ATP supplies energy to cells in order to synthesise large molecules, transporting molecules through active transport and moving material around the cell. Cells create ATP through cell respiration - the energy that is not converted into ATP is converted into heat. When molecules are digested or used for activities they also release heat energy.
U4.2.5 Living organisms cannot convert heat to other forms of energy.
Living organisms can perform various energy conversions:
- Light energy to chemical energy in photosynthesis
- Chemical energy to kinetic energy in muscle contraction
- Chemical energy to electrical energy in nerve cells
- Chemical energy to heat energy in heat-generating adipose tissue.
They cannot convert heat energy into any other form of energy.
U4.2.6 Heat is lost from ecosystems.
Heat resulting from cell respiration makes living organisms warmer. This heat can be useful in making cold-blooded animals more active. Birds and mammals increase their rate of heat generation. if necessary to maintain there constant body temperatures.
According to the laws of thermodynamics in physics, heat passes from hotter to cooler bodies, so heat produced in living organisms is all eventually lost to the abiotic environment. The heat may remain in the ecosystem for a while, but ultimately is lost, for example when heat is radiated into the atmosphere.
U4.2.7 Energy losses between trophic levels restrict the length of food chains and the biomass of higher trophic levels.
Biomass is the total mass of a group of living or recently living organisms. It consists of the cells and tissues of those organisms, including the carbohydrates and other carbon compounds that they contain. Because carbon compounds have chemical energy, biomass has energy.
Ecologists can measure how much energy is added per year by groups of organisms to their biomass. The results are calculated per square meter of the ecosystem so that different trophic levels can be compared. When this is done, the same trend is always found: the energy added to biomass by each successive trophic level is less. In secondary consumers, for example, the amount of energy is always less per year per square meter of ecosystem than in primary consumers.
Because of these losses, only a small proportion of the energy in the biomass of organisms in one trophic level will ever become part of the biomass of organisms in the next trophic level. As the losses occur at each stage in a food chain, there is less and less energy available to each successive trophic level. After only a few stages in a food chain the amount of energy remaining would not be enough to support another trophic level. Biomass, measured in grams, also diminishes along food chains, due to loss of carbon dioxide and water from respiration and loss form the food chain of uneaten or undigested parts of organisms.
APPLICATION:
N/A
SKILL:
S4.2.1 Quantitative representations of energy flow using pyramids of energy.
UNDERSTANDINGS:
U4.2.1 Most ecosystems rely on a supply of energy from sunlight.
The most important organisms in any food chain are the producers because without them, the next trophic level would have nothing to consume. Since photosynthetic organisms occupy the first trophic level, sunlight is the initial energy source for almost all communities.
The vast majority of the foods we eat,the energy they contain can be traced back to sunlight. Milk, for example, gets its energy from the cow which produced it. The cow obtained its energy from grass, which got its energy from sunlight. In marine ecosystems, most food chains start with phytoplankton which get their energy from sunlight.
Leaves are a food source when they are green and photosynthesising and continue to be a source of food when they have fallen to the ground and turned brown. Decomposers and detritus feeders such as soil organisms or some of the bottom feeders in a river depend on this dead organic matter for food.
Sunlight is almost always the first type of energy to enter an ecosystem. Some living things, like plants, are able to capture the energy from sunlight. When another living thing in an ecosystem eats a plant, it is gaining energy that came first from the sun.
U4.2.2 Light energy is converted to chemical energy in carbon compounds by photosynthesis.
Photosynthesis is a process used by plants and other organisms to convert light energy, normally from the sun, into chemical energy that can be later released to fuel the organisms' activities. This chemical energy is stored in carbohydrate molecules, such as sugars, which are synthesised from carbon dioxide and water.
Producers absorb light energy through photosynthetic pigments which convert it to chemical energy. This chemical energy is used to create carbon compounds such as carbohydrates and lipids. These producers release energy from their carbon compounds by cell respiration, the rest remains in their cells and tissues to provide it to the heterotrophs
U4.2.3 Chemical energy in carbon compounds flows through food chains by means of feeding.
Producers are the first organisms in a food chain since they obtain energy from sunlight instead of from other organisms. Organisms that feed on producers and obtain energy from them are primary consumers in the food chain. Organisms that feed on primary consumers are secondary consumers. Organisms that feed on secondary consumers are tertiary consumers (and so on). The arrows on a food chain show the transportation of energy, which is why an arrow flows from the first organisms (producers/primary consumers) to the last (secondary/tertiary consumers).
U4.2.4 Energy released from carbon compounds by respiration is used in living organisms and converted to heat.
Producers can release energy from their carbon compounds by cell respiration and then use it for cell activities. Energy released in this way is eventually lost to the environment as waste heat. However, only some of the carbon compounds in producers are used in this way and the largest part remains in the cells and tissues of producers. The energy in these carbon compounds is available to heterotrophs.
ATP supplies energy to cells in order to synthesise large molecules, transporting molecules through active transport and moving material around the cell. Cells create ATP through cell respiration - the energy that is not converted into ATP is converted into heat. When molecules are digested or used for activities they also release heat energy.
U4.2.5 Living organisms cannot convert heat to other forms of energy.
Living organisms can perform various energy conversions:
- Light energy to chemical energy in photosynthesis
- Chemical energy to kinetic energy in muscle contraction
- Chemical energy to electrical energy in nerve cells
- Chemical energy to heat energy in heat-generating adipose tissue.
They cannot convert heat energy into any other form of energy.
U4.2.6 Heat is lost from ecosystems.
Heat resulting from cell respiration makes living organisms warmer. This heat can be useful in making cold-blooded animals more active. Birds and mammals increase their rate of heat generation. if necessary to maintain there constant body temperatures.
According to the laws of thermodynamics in physics, heat passes from hotter to cooler bodies, so heat produced in living organisms is all eventually lost to the abiotic environment. The heat may remain in the ecosystem for a while, but ultimately is lost, for example when heat is radiated into the atmosphere.
U4.2.7 Energy losses between trophic levels restrict the length of food chains and the biomass of higher trophic levels.
Biomass is the total mass of a group of living or recently living organisms. It consists of the cells and tissues of those organisms, including the carbohydrates and other carbon compounds that they contain. Because carbon compounds have chemical energy, biomass has energy.
Ecologists can measure how much energy is added per year by groups of organisms to their biomass. The results are calculated per square meter of the ecosystem so that different trophic levels can be compared. When this is done, the same trend is always found: the energy added to biomass by each successive trophic level is less. In secondary consumers, for example, the amount of energy is always less per year per square meter of ecosystem than in primary consumers.
Because of these losses, only a small proportion of the energy in the biomass of organisms in one trophic level will ever become part of the biomass of organisms in the next trophic level. As the losses occur at each stage in a food chain, there is less and less energy available to each successive trophic level. After only a few stages in a food chain the amount of energy remaining would not be enough to support another trophic level. Biomass, measured in grams, also diminishes along food chains, due to loss of carbon dioxide and water from respiration and loss form the food chain of uneaten or undigested parts of organisms.
APPLICATION:
N/A
SKILL:
S4.2.1 Quantitative representations of energy flow using pyramids of energy.
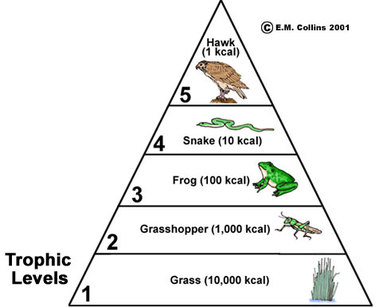
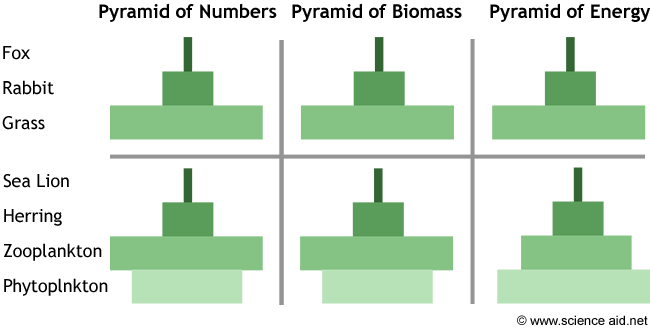
- A pyramid of energy is a graphical representation of the amount of energy of each tropic level in a food chain
- They are expressed in units of energy per area per time (e.g. kJ m2 year -1)
- Pyramids of energy will never appear inverted as some of the energy stored in one source is always lost when transferred to the next source
- This is an application of the second law of thermodynamics
- Each level of the pyramid of energy should be approximately one tenth the size of the level preceding it, as energy transformations are ~10% efficient
Essential idea: Continued availability of carbon in ecosystems depends on carbon cycling.
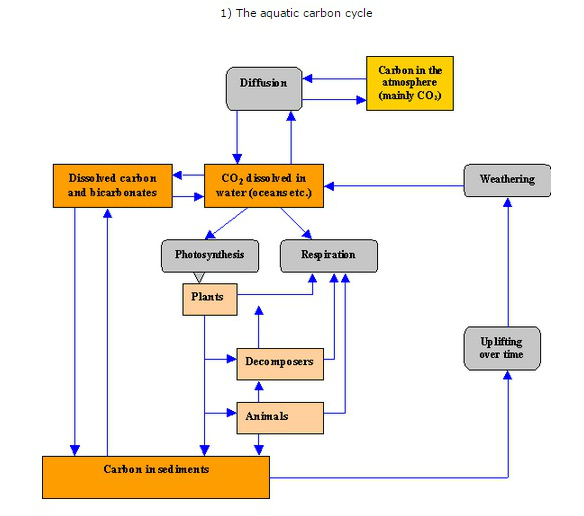
4.3 Carbon cycling
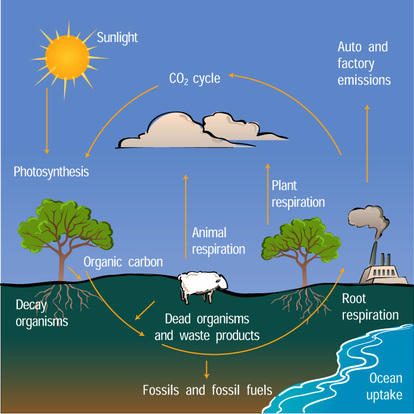
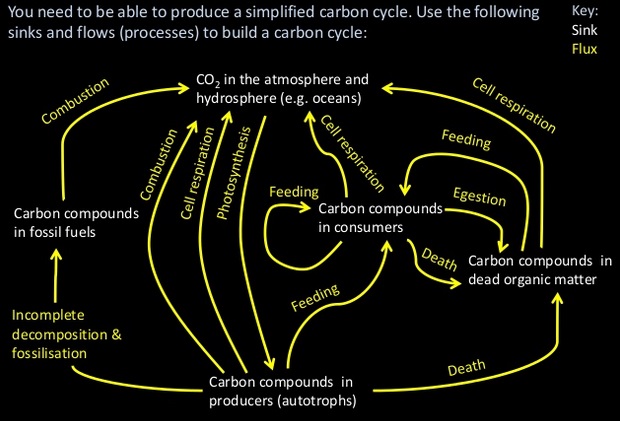
U4.3.1 Autotrophs convert carbon dioxide into carbohydrates and other carbon compounds.
All autotrophs convert carbon dioxide, from the atmosphere or dissolved in water, into organic compounds. Plants initially synthesise sugars e.g. glucose which are then converted into other organic compounds such as complex carbohydrates - starch and cellulose, lipids and amino acids.
U4.3.2 In aquatic ecosystems carbon is present as dissolved carbon dioxide and hydrogencarbonate ions.
U4.3.3 Carbon dioxide diffuses from the atmosphere or water into autotrophs.
U4.3.4 Carbon dioxide is produced by respiration and diffuses out of organisms into water or the atmosphere.
There is a constant exchange between the upper layers of the oceans and the atmosphere. Nature strives towards equilibrium, and thus for the ocean and the atmosphere to contain equal concentrations of CO2. Carbon dioxide in the atmosphere therefore dissolves in the surface waters of the oceans in order to establish a concentration in equilibrium with that of the atmosphere. As CO2 dissolves in the ocean it generates dramatic changes in sea water chemistry. CO2 reacts with water molecules (H2O) and forms the weak acid H2CO3 (carbonic acid). Most of this acid dissociates into hydrogen ions (H+) and bicarbonate ions (HCO3-).
U4.3.5 Methane is produced from organic matter in anaerobic conditions by methanogenic archaeans and some diffuses into the atmosphere or accumulates in the ground.
Methanogens are archaean (old) microorganisms that produce methane as a metabolic byproduct in anoxic* conditions. Typically during ATP production, methane is produced from carbon dioxide.
Methanogens are found in a variety of anoxic conditions:
* Anaerobic = total absence of free oxygen (O2) or bound oxygen (NO2, NO3)
Anoxic = absence of free oxygen, but presence of bound oxygen.
U4.3.6 Methane is oxidized to carbon dioxide and water in the atmosphere.
Methane has both natural and anthropogenic sources. It is produced wherever organic matter decays under conditions where oxygen is lacking. This is the case, for example, in wet rice fields, in waste deposits, and in the stomachs of cattle and of termites. Also, methane is released while obtaining it from underground reservoirs for industrial use, while pumping oil, and while mining coal.
A substantial amount of methane apparently also originates from the burning of forests and other vegetation ("biomass" burning). Methane is relatively short-lived in the atmosphere; a molecule of methane is oxidized to water and carbon dioxide within a decade or so, mainly by reaction with another trace gas, the hydroxyl radical OH-.
Thus, unlike the case of carbon dioxide (which stays in the atmosphere longer than methane), a concerted effort to reduce methane emissions would have almost immediate results in terms of reduction of greenhouse effect. As in the case of carbon dioxide, the origin of methane can be partly traced by recording the amount of radiocarbon in the methane (fossil fuels have none) and by measuring the ratio of carbon-13 to carbon-12 (bacteria produce methane high in 12C).
U4.3.7 Peat forms when organic matter is not fully decomposed because of acidic and/or anaerobic conditions in waterlogged soils.
U4.3.1 Autotrophs convert carbon dioxide into carbohydrates and other carbon compounds.
All autotrophs convert carbon dioxide, from the atmosphere or dissolved in water, into organic compounds. Plants initially synthesise sugars e.g. glucose which are then converted into other organic compounds such as complex carbohydrates - starch and cellulose, lipids and amino acids.
U4.3.2 In aquatic ecosystems carbon is present as dissolved carbon dioxide and hydrogencarbonate ions.
U4.3.3 Carbon dioxide diffuses from the atmosphere or water into autotrophs.
U4.3.4 Carbon dioxide is produced by respiration and diffuses out of organisms into water or the atmosphere.
There is a constant exchange between the upper layers of the oceans and the atmosphere. Nature strives towards equilibrium, and thus for the ocean and the atmosphere to contain equal concentrations of CO2. Carbon dioxide in the atmosphere therefore dissolves in the surface waters of the oceans in order to establish a concentration in equilibrium with that of the atmosphere. As CO2 dissolves in the ocean it generates dramatic changes in sea water chemistry. CO2 reacts with water molecules (H2O) and forms the weak acid H2CO3 (carbonic acid). Most of this acid dissociates into hydrogen ions (H+) and bicarbonate ions (HCO3-).
U4.3.5 Methane is produced from organic matter in anaerobic conditions by methanogenic archaeans and some diffuses into the atmosphere or accumulates in the ground.
Methanogens are archaean (old) microorganisms that produce methane as a metabolic byproduct in anoxic* conditions. Typically during ATP production, methane is produced from carbon dioxide.
Methanogens are found in a variety of anoxic conditions:
- Wetlands (paddies, swamps and mangroves)
- Digestive tracts of animals (cows, humans and termites)
- Marine and freshwater sediments (mud in the beds of lakes)
- Landfill sites (in which organic matter has been buried).
* Anaerobic = total absence of free oxygen (O2) or bound oxygen (NO2, NO3)
Anoxic = absence of free oxygen, but presence of bound oxygen.
U4.3.6 Methane is oxidized to carbon dioxide and water in the atmosphere.
Methane has both natural and anthropogenic sources. It is produced wherever organic matter decays under conditions where oxygen is lacking. This is the case, for example, in wet rice fields, in waste deposits, and in the stomachs of cattle and of termites. Also, methane is released while obtaining it from underground reservoirs for industrial use, while pumping oil, and while mining coal.
A substantial amount of methane apparently also originates from the burning of forests and other vegetation ("biomass" burning). Methane is relatively short-lived in the atmosphere; a molecule of methane is oxidized to water and carbon dioxide within a decade or so, mainly by reaction with another trace gas, the hydroxyl radical OH-.
Thus, unlike the case of carbon dioxide (which stays in the atmosphere longer than methane), a concerted effort to reduce methane emissions would have almost immediate results in terms of reduction of greenhouse effect. As in the case of carbon dioxide, the origin of methane can be partly traced by recording the amount of radiocarbon in the methane (fossil fuels have none) and by measuring the ratio of carbon-13 to carbon-12 (bacteria produce methane high in 12C).
U4.3.7 Peat forms when organic matter is not fully decomposed because of acidic and/or anaerobic conditions in waterlogged soils.

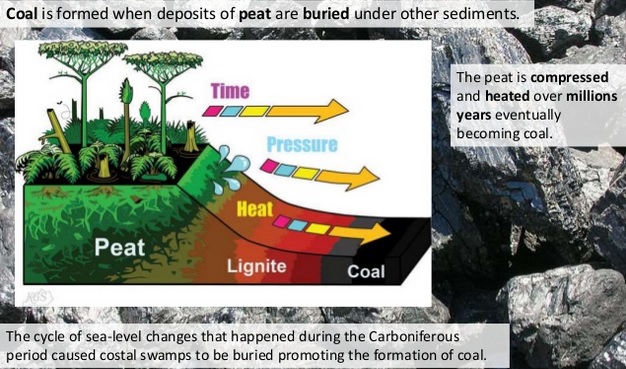
U4.3.8 Partially decomposed organic matter from past geological eras was converted either into coal or into oil and gas that accumulate in porous rocks.
Also, oil and gas formation occurred in ancient oceans:
Tiny sea plants and animals died and were buried on the ocean floor. Over time, they were covered by layers of silt and sand (conditions were/are anaerobic so decomposition was only partial).
Over a long period of time, the remains were buried deeper and deeper. The enormous heat and pressure turned them into oil and and gas. The mixture of different types of oil and gas is the result of complex chemical changes. Methane forms the largest part of natural gas.
Also, oil and gas formation occurred in ancient oceans:
Tiny sea plants and animals died and were buried on the ocean floor. Over time, they were covered by layers of silt and sand (conditions were/are anaerobic so decomposition was only partial).
Over a long period of time, the remains were buried deeper and deeper. The enormous heat and pressure turned them into oil and and gas. The mixture of different types of oil and gas is the result of complex chemical changes. Methane forms the largest part of natural gas.
U4.3.9 Carbon dioxide is produced by the combustion of biomass and fossilized organic matter.
Carbon can be stored as biomass in the roots of trees and other organic matter for many decades. This carbon is released back into the atmosphere by decomposition. Not all organic matter is immediately decomposed. Under certain conditions dead plant matter accumulates faster than it is decomposed within an ecosystem. The remains are locked away in underground deposits.
When layers of sediment compress this matter fossil fuels will be formed, after many centuries. Long-term geological processes may expose the carbon in these fuels to air after a long period of time, but usually the carbon within the fossil fuels is released during humane combustion processes.
The combustion of fossil fuels has supplied us with energy for as long as we can remember. But the human population of the world has been expanding and so has our demand for energy. That is why fossil fuels are burned very extensively. This is not without consequences, because we are burning fossil fuels much faster than they develop. Because of our actions fossil fuels have become non-renewable recourses.
U4.3.10 Animals such as reef-building corals and mollusca have hard parts that are composed of calcium carbonate and can become fossilized in limestone.
Some animals secrete calcium carbonate structures to protect themselves e.g. shells of molluscs and hard coral exoskeletons. When the animals die, the soft body parts decompose but the calcium carbonate remains to form deposits on the ocean floor. The deposits are buried and compressed, eventually forming limestone rock. Imprints of the hard body parts remain in the rock as fossils.
NOTE: limestone rock is a huge carbon sink.
APPLICATION:
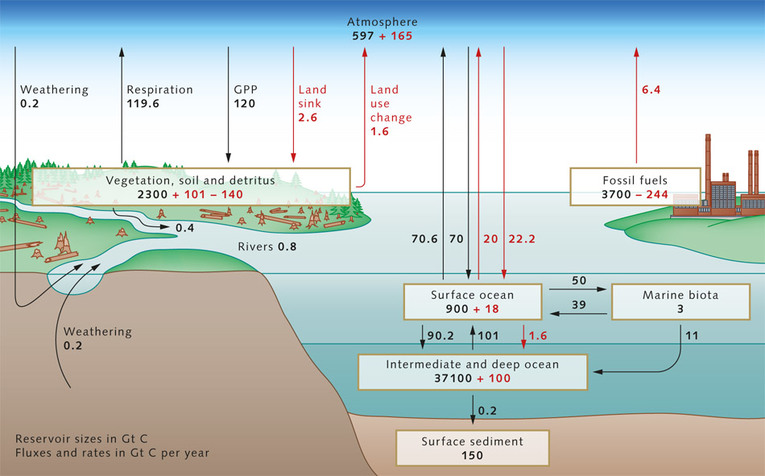
A4.3.1 Estimation of carbon fluxes due to processes in the carbon cycle.
Carbon can be stored as biomass in the roots of trees and other organic matter for many decades. This carbon is released back into the atmosphere by decomposition. Not all organic matter is immediately decomposed. Under certain conditions dead plant matter accumulates faster than it is decomposed within an ecosystem. The remains are locked away in underground deposits.
When layers of sediment compress this matter fossil fuels will be formed, after many centuries. Long-term geological processes may expose the carbon in these fuels to air after a long period of time, but usually the carbon within the fossil fuels is released during humane combustion processes.
The combustion of fossil fuels has supplied us with energy for as long as we can remember. But the human population of the world has been expanding and so has our demand for energy. That is why fossil fuels are burned very extensively. This is not without consequences, because we are burning fossil fuels much faster than they develop. Because of our actions fossil fuels have become non-renewable recourses.
U4.3.10 Animals such as reef-building corals and mollusca have hard parts that are composed of calcium carbonate and can become fossilized in limestone.
Some animals secrete calcium carbonate structures to protect themselves e.g. shells of molluscs and hard coral exoskeletons. When the animals die, the soft body parts decompose but the calcium carbonate remains to form deposits on the ocean floor. The deposits are buried and compressed, eventually forming limestone rock. Imprints of the hard body parts remain in the rock as fossils.
NOTE: limestone rock is a huge carbon sink.
APPLICATION:
A4.3.1 Estimation of carbon fluxes due to processes in the carbon cycle.
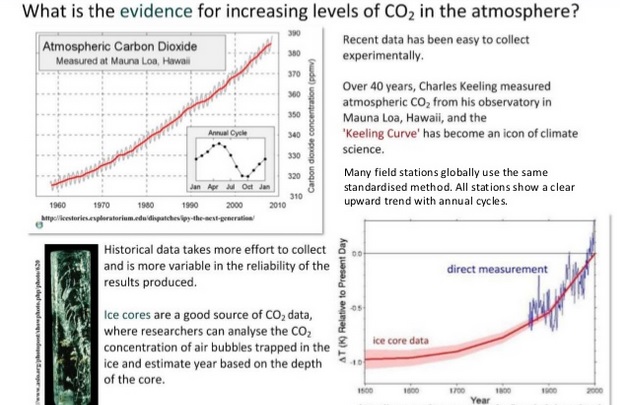
A4.3.2 Analysis of data from air monitoring stations to explain
annual fluctuations.
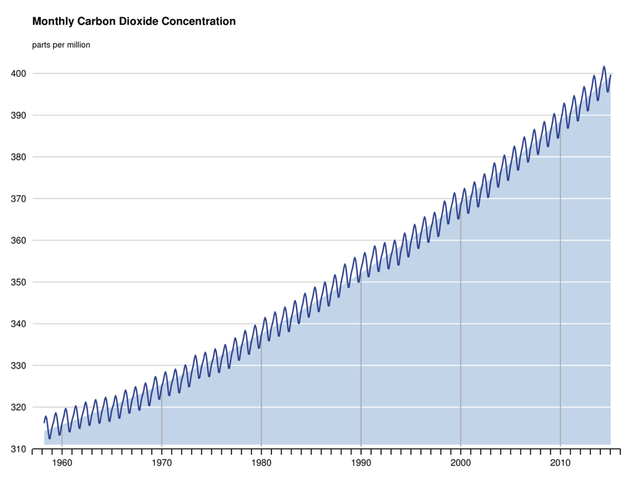
Monthly carbon dioxide concentrations at NOAA's Mauna Loa Observatory. This graph shows an annual seasonal cycle and a steady upward trend since CO2 measurements began atop Mauna Loa, Hawaii, in 1958.
The seasonal cycle is due to the vast land mass of the Northern Hemisphere, which contains the majority of land-based vegetation. The result is a decrease in atmospheric carbon dioxide during northern spring and summer, when plants are absorbing CO2 as part of photosynthesis. The pattern reverses, with an increase in atmospheric carbon dioxide during northern fall and winter. The yearly spikes during the cold months occur as annual vegetation dies and leaves fall and decompose, which releases their carbon back into the air.
This graphic portrayal of rising CO2 levels is known as the Keeling curve in honor of the originator of these measurements, Charles David Keeling of the Scripps Institution of Oceanography.
Essential idea: Concentrations of gases in the atmosphere affect climates experienced at the Earth’s surface.
4.4 Climate change
UNDERSTANDINGS:
U4.4.1 Carbon dioxide and water vapour are the most significant greenhouse gases.
U4.4.2 Other gases including methane and nitrogen oxides have less impact.
U4.4.3 The impact of a gas depends on its ability to absorb long wave radiation as well as on its concentration in the atmosphere.
U4.4.4 The warmed Earth emits longer wavelength radiation (heat).
U4.4.5 Longer wave radiation is absorbed by greenhouse gases that retain the heat in the atmosphere.
U4.4.6 Global temperatures and climate patterns are influenced by concentrations of greenhouse gases.
Unlike CO2, which can persist in the air for centuries, water vapour cycles through the atmosphere quickly, evaporating from the oceans and elsewhere before coming back down as rain or snow. Since the rate of evaporation rises with temperature, the amount of water vapour in the air at any one time (and the amount of warming it causes) is strongly related to the amount of other greenhouse gases in the air.
Water vapor is the most abundant heat-trapping gas, but rarely discussed when considering human-induced climate change. The principal reason is that water vapor has a short cycle in the atmosphere (a few days) before it is incorporated into weather events and falls to Earth, so it cannot build up in the atmosphere in the same way as carbon dioxide does.
Long-wave radiation absorbed by these gases in turn is re-emitted in all directions, including back toward Earth and some of this re-emitted energy is absorbed again by these gases and re-emitted in all directions. The net effect is that most of the outgoing radiation is kept within the atmosphere instead of escaping into space. Heat-trapping gases, in balanced proportions, act like a blanket surrounding Earth, keeping temperatures within a range that enables life to thrive on a planet with liquid water. Unfortunately, these gases—especially carbon dioxide—are accumulating in the atmosphere at increasing concentrations due to human activities such as the burning of fossil fuel in cars and power plants, the clearing of forests for agriculture or development, and agricultural practices. As a result, the insulating blanket is getting too thick and overheating the Earth as less energy (heat) escapes into space.
Arguments AGAINST human activity being the cause for global warming:
UNDERSTANDINGS:
U4.4.1 Carbon dioxide and water vapour are the most significant greenhouse gases.
U4.4.2 Other gases including methane and nitrogen oxides have less impact.
U4.4.3 The impact of a gas depends on its ability to absorb long wave radiation as well as on its concentration in the atmosphere.
U4.4.4 The warmed Earth emits longer wavelength radiation (heat).
U4.4.5 Longer wave radiation is absorbed by greenhouse gases that retain the heat in the atmosphere.
U4.4.6 Global temperatures and climate patterns are influenced by concentrations of greenhouse gases.
- Carbon dioxide (CO2) : Carbon dioxide enters the atmosphere through burning fossil fuels (coal, natural gas and oil), solid waste, trees and wood products, and also as a result of certain chemical reactions (e.g. manufacture of cement). Carbon dioxide is removed from the atmosphere (or "sequestered") when it is absorbed by plants as part of the biological carbon cycle.
- Methane (CH4) : Methane is emitted during the production and transport of coal, natural gas, and oil. Methane emissions also result from livestock and other agricultural practices and by the decay of organic waste in municipal solid waste landfills.
- Nitrous oxide (N2O) : Nitrous oxide is emitted during agricultural and industrial activities, as well as during combustion of fossil fuels and solid waste.
- Fluorinated gases : Hydrofluorocarbons, perfluorocarbons, sulfur hexafluoride, and nitrogen trifluoride are synthetic, powerful greenhouse gases that are emitted from a variety of industrial processes. Fluorinated gases are sometimes used as substitutes for stratospheric ozone-depleting substances (e.g. chlorofluorocarbons, hydrochlorofluorocarbons, and halons). These gases are typically emitted in smaller quantities, but they are potent greenhouse gases.
Unlike CO2, which can persist in the air for centuries, water vapour cycles through the atmosphere quickly, evaporating from the oceans and elsewhere before coming back down as rain or snow. Since the rate of evaporation rises with temperature, the amount of water vapour in the air at any one time (and the amount of warming it causes) is strongly related to the amount of other greenhouse gases in the air.
Water vapor is the most abundant heat-trapping gas, but rarely discussed when considering human-induced climate change. The principal reason is that water vapor has a short cycle in the atmosphere (a few days) before it is incorporated into weather events and falls to Earth, so it cannot build up in the atmosphere in the same way as carbon dioxide does.
Long-wave radiation absorbed by these gases in turn is re-emitted in all directions, including back toward Earth and some of this re-emitted energy is absorbed again by these gases and re-emitted in all directions. The net effect is that most of the outgoing radiation is kept within the atmosphere instead of escaping into space. Heat-trapping gases, in balanced proportions, act like a blanket surrounding Earth, keeping temperatures within a range that enables life to thrive on a planet with liquid water. Unfortunately, these gases—especially carbon dioxide—are accumulating in the atmosphere at increasing concentrations due to human activities such as the burning of fossil fuel in cars and power plants, the clearing of forests for agriculture or development, and agricultural practices. As a result, the insulating blanket is getting too thick and overheating the Earth as less energy (heat) escapes into space.
Arguments AGAINST human activity being the cause for global warming:
- The Earth’s orbit around the Sun is an ellipse, not a circle but the ellipse changes shape. Sometimes it is almost circular and the Earth stays approximately the same distance from the Sun as it progresses around its orbit. At other times the ellipse is more pronounced so that the Earth moves closer and further away from the sun as it orbits. When the Earth is closer to the sun our climate is warmer because the sun is expanding.
- The Earth rotates around an axis (imagine a line that joins the north and south poles) but the Earth’s axis is not upright, it leans at an angle. This angle changes with time and over about 41 000 years it moves from 22.1 degrees to 24.5 degrees and back again. When the angle increases the summers become warmer and the winters become colder. The earth naturally fluctuates in temperature. we just happen to be on the ascending limb of the fluctuation.
- Human releases of CO2 cannot cause climate change as any increases in CO2 are eventually balanced by nature. CO2 gets absorbed by oceans, forests, and other "carbon sinks" that increase their biological activity to absorb excess CO2 from the atmosphere. 50% of the CO2 released by the burning of fossil fuels and other human activities, has already been absorbed.
- Changes in ocean currents are primarily responsible for the melting Greenland ice sheet, Arctic sea ice, and Arctic permafrost. Over the 20th century there have been two Arctic warming periods with a cooling period (1940-1970) in between. According to a peer-reviewed Apr. 19, 2009 study in Geophysical Research Letters, natural shifts in the ocean currents are the major cause of these climate changes, not human generated greenhouse gases.
- The increased hurricane activity over the past decade (1995-2005), including hurricane Katrina, is not the result of human-induced climate change; it is the result of cyclical tropical cyclone patterns, driven primarily by natural ocean currents, according to the National Oceanic and Atmospheric Administration's (NOAA) testimony in the US Senate on Sep. 20, 2005.
- Global warming and cooling are caused by fluctuations in the sun's heat (solar forcing), not by the minor greenhouse effect of human-produced gases such as CO2 and methane (CH4). Between 1900 and 2000 solar irradiance increased .19%. This increase correlates with the rise in surface temperatures in the US.