Essential idea: Physical and chemical properties depend on the ways in which different atoms combine.
1.1 Introduction to the particulate nature of matter and chemical change
UNDERSTANDINGS:
U1.1.1 Atoms of different elements combine in fixed ratios to form compounds, which have different properties from their component elements.
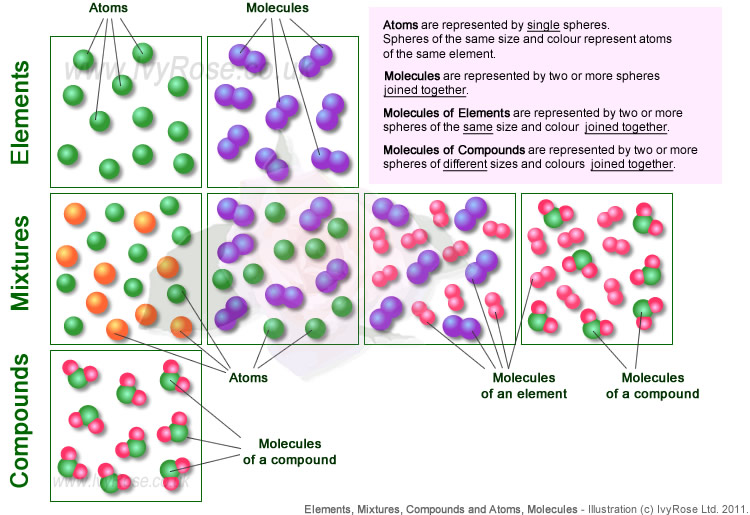
A compound is the result of two or more elements becoming chemically combined together. The elements react chemically, causing bonds to be formed between the atoms involved ... resulting in molecules of the compound. The elements react in specific amounts, resulting in a compound that has a specific chemical composition. Compounds do not necessarily resemble the elements of which they are composed and cannot be converted back to those elements by physical methods. It can even be very difficult and require a lot of energy to change compounds into their elements by chemical means (reactions).
A compound is a pure substance that consists of two or more elements chemically combined in a fixed proportion, that can be further subdivided into simpler substances by chemical (not physical) means only. ... So, if a quantity of a material consists of atoms of two or more elements joined together, always in the same ratio, then the matter forming that material is a compound.
U1.1.2 Mixtures contain more than one element and/or compound that are not chemically bonded together and so retain their individual properties.
U1.1.3 Mixtures are either homogeneous or heterogeneous.
UNDERSTANDINGS:
U1.1.1 Atoms of different elements combine in fixed ratios to form compounds, which have different properties from their component elements.
A compound is the result of two or more elements becoming chemically combined together. The elements react chemically, causing bonds to be formed between the atoms involved ... resulting in molecules of the compound. The elements react in specific amounts, resulting in a compound that has a specific chemical composition. Compounds do not necessarily resemble the elements of which they are composed and cannot be converted back to those elements by physical methods. It can even be very difficult and require a lot of energy to change compounds into their elements by chemical means (reactions).
A compound is a pure substance that consists of two or more elements chemically combined in a fixed proportion, that can be further subdivided into simpler substances by chemical (not physical) means only. ... So, if a quantity of a material consists of atoms of two or more elements joined together, always in the same ratio, then the matter forming that material is a compound.
- Compounds consist of molecules formed from atoms of 2 or more different elements bound together chemically.
- Compounds can be broken down into a simpler type of matter (elements) by chemical means; not by physical means
- Compounds always contains the same ratio of component elements.
- Compounds have properties different from their component elements
e.g. the compound water (H2O) is a liquid at room temperature and pressure and has different chemical properties from those of the two elements, hydrogen (H2) and oxygen (O2), from which it is formed. - Compounds can be represented using chemical formulae.
U1.1.2 Mixtures contain more than one element and/or compound that are not chemically bonded together and so retain their individual properties.
U1.1.3 Mixtures are either homogeneous or heterogeneous.
- Consist of two or more different elements and/or compounds - physically intermingled i.e. not chemically joined.
- Can be separated into their constituent parts by physical means (e.g. distilation of liquids or seperating magnetic and non-magnetic solids using a magnet)
- Have many of the properties of their constituent parts (e.g. the element "oxygen" is part of the mixture "air" and some of the properties of air are due to the oxygen, albeit somewhat reduced compared with pure oxygen due to the presence of the other constituents of the mixture called "air").
- Homogeneous Mixtures - in which the two or more substances that form the mixture are evenly distributed throughout the mixture i.e. has uniform composition and properties throughout, e.g. vinegar is a homogeneous mixture of ethanoic acid and water.
- Heterogeneous Mixtures - in which the two or more substances that form the mixture are not evenly distributed throughout the mixture i.e. has non-uniform composition and varying properties, e.g. oil and water.
- Solutions - a special type of homogeneous mixtures in which one substance (called the "solute") is dissolved in another substance (called the "solvent"), e.g. salt water is salt dissolved in water - in such a way that the salt no longer exists as solid particles within the water.
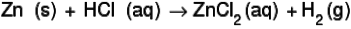
APPLICATION:
A1.1.1 Application of the state symbols (s), (l), (g) and (aq) in equations.
s = solid
l = liquid
g = gas
aq = aqueous
NOTE: A common question asked is how to differentiate between a liquid and an aqueous solution:
A liquid is a pure element or compound e.g. water.
An aqueous solution is where water is the solvent in the solution e.g. aqueous HCl is hydrochloric acid dissolved in water.
SKILL:
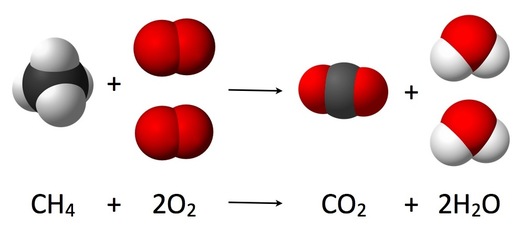
S1.1.1 Deduction of chemical equations when reactants and products are specified.
Reactants = methane and oxygen
Products = carbon dioxide and water
A1.1.1 Application of the state symbols (s), (l), (g) and (aq) in equations.
s = solid
l = liquid
g = gas
aq = aqueous
NOTE: A common question asked is how to differentiate between a liquid and an aqueous solution:
A liquid is a pure element or compound e.g. water.
An aqueous solution is where water is the solvent in the solution e.g. aqueous HCl is hydrochloric acid dissolved in water.
SKILL:
S1.1.1 Deduction of chemical equations when reactants and products are specified.
Reactants = methane and oxygen
Products = carbon dioxide and water
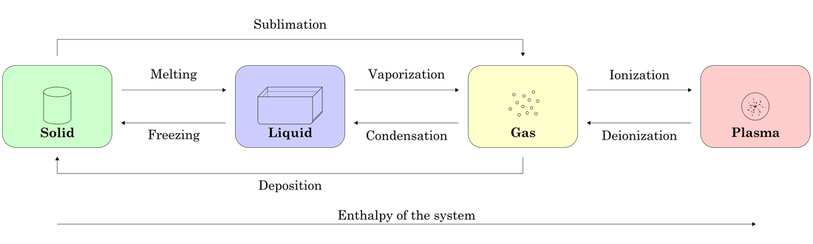
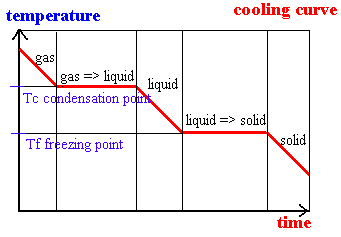
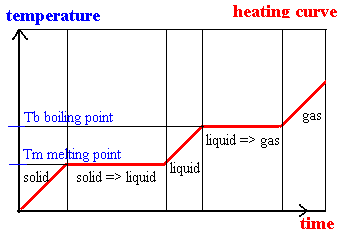
S1.1.2 Explanation of observable changes in physical properties and temperature during changes of state.
HEATING CURVE ANALYSIS - e.g. ICE to STEAM:
Point A = beginning of solid up-slope
Point B = top of solid up-slope
Point C = start of solid-liquid straight line
Point D = end of solid-liquid straight line
Point E = midpoint of liquid up-slope
Point F = start of liquid-gas straight line
Point G = beginning of gas up-slope
Point H = midpoint of gas up-slope
A -> B = ice rises in temperature from -10 degrees to 0 degrees (still solid) - gain in thermal energy
C -> D = ice melts at 0 degrees - no rise in temperature as heat energy is used up for bond breaking - particles are solid/liquid
D -> E = liquid water rises in temperature from 0 degrees to 100 degrees - water exists as a liquid (completely)
F -> G = liquid water boils at 100 degrees - evaporation (vapourisation) - no rise in temperature as energy is used to break bonds - paarticles are liquid/gas
G -> H = steam rises in temperature from 100 degrees to 120 degrees - particles are completely in gas form and all bonds are broken.
Point A = beginning of solid up-slope
Point B = top of solid up-slope
Point C = start of solid-liquid straight line
Point D = end of solid-liquid straight line
Point E = midpoint of liquid up-slope
Point F = start of liquid-gas straight line
Point G = beginning of gas up-slope
Point H = midpoint of gas up-slope
A -> B = ice rises in temperature from -10 degrees to 0 degrees (still solid) - gain in thermal energy
C -> D = ice melts at 0 degrees - no rise in temperature as heat energy is used up for bond breaking - particles are solid/liquid
D -> E = liquid water rises in temperature from 0 degrees to 100 degrees - water exists as a liquid (completely)
F -> G = liquid water boils at 100 degrees - evaporation (vapourisation) - no rise in temperature as energy is used to break bonds - paarticles are liquid/gas
G -> H = steam rises in temperature from 100 degrees to 120 degrees - particles are completely in gas form and all bonds are broken.
The quantity of heat absorbed or released by a substance undergoing a change of state, such as ice changing to water or water to steam, at constant temperature and pressure is known as latent heat. This definition is useful to know however is not required as part of the 2016 IB syllabus.
Essential idea: The mole makes it possible to correlate the number of particles with the mass that can be measured.
1.2 The mole concept
UNDERSTANDINGS:
U1.2.1 The mole is a fixed number of particles and refers to the amount, n, of substance.
U1.2.3 Molar mass (M) has the units g mol-1.
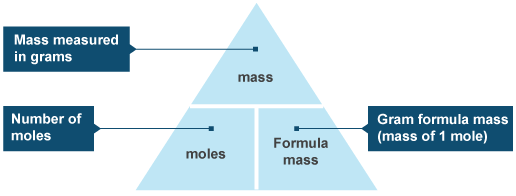
A mole is a convenient way of counting amounts of substances in chemistry. Because atoms are so miniscule, it's useless (and basically impossible) to count them individually, so we use a number called the mole as a unit. The number below is know as Avogadro's number.
1 mole is 602,000,000,000,000,000,000,000 particles (6.02 x 10^23). These particles could be atoms, electrons, ions, molecules etc.
ONE MOLE OF A SUBSTANCE = THE FORMULA MASS OF THE SUBSTANCE IN GRAMS
For example, 1 mole of carbon has a mass of 12g because the relative formula mass (relative atomic mass in this case since carbon is a non-diatomic element) of carbon is 12.
1 mole of water has a mass of 18g.
2 moles of water has a mass of 36g.
As shown by the diagram above, moles (n) = mass (g) ÷ Mr (relative formula mass on periodic table - g/mol)
For example:
How many moles are there in 100g of NaOH(s)?
n = m ÷ Mr
= 100 ÷ 40
= 2.5 mol
UNDERSTANDINGS:
U1.2.1 The mole is a fixed number of particles and refers to the amount, n, of substance.
U1.2.3 Molar mass (M) has the units g mol-1.
A mole is a convenient way of counting amounts of substances in chemistry. Because atoms are so miniscule, it's useless (and basically impossible) to count them individually, so we use a number called the mole as a unit. The number below is know as Avogadro's number.
1 mole is 602,000,000,000,000,000,000,000 particles (6.02 x 10^23). These particles could be atoms, electrons, ions, molecules etc.
ONE MOLE OF A SUBSTANCE = THE FORMULA MASS OF THE SUBSTANCE IN GRAMS
For example, 1 mole of carbon has a mass of 12g because the relative formula mass (relative atomic mass in this case since carbon is a non-diatomic element) of carbon is 12.
1 mole of water has a mass of 18g.
2 moles of water has a mass of 36g.
As shown by the diagram above, moles (n) = mass (g) ÷ Mr (relative formula mass on periodic table - g/mol)
For example:
How many moles are there in 100g of NaOH(s)?
n = m ÷ Mr
= 100 ÷ 40
= 2.5 mol
U1.2.2 Masses of atoms are compared on a scale relative to 12C and are expressed as relative atomic mass (A r) and relative formula/molecular mass (M r).
Chemists don't deal with the masses of atoms and molecules measured in grams because they would be dealing with extremely small numbers. Instead, the masses of atoms and molecules are compared with that of the isotope carbon-12. 12C is assigned a relative atomic mass of 12. As an example, consider an atom of magnesium. Two atoms of 12C are required to be equal in mass to one atom of Mg therefore the relative atomic mass of Mg is 24.
U1.2.4 The empirical formula and molecular formula of a compound give the simplest ratio and the actual number of atoms present in a molecule respectively.
(Application and Skill) AS1.2.3 Interconversion of the percentage composition by mass and the empirical formula.
AS1.2.4 Determination of the molecular formula of a compound from its empirical formula and molar mass.
Example 1:
A compound is found to contain 50.05 % sulfur and 49.95 % oxygen by weight. What is the empirical formula for this compound? The molecular weight for this compound is 64.07 g/mol. What is its molecular formula?
Solution:
1) Assume 100 g of the compound is present. This changes the percents to grams:
S ⇒ 50.05 g
O ⇒ 49.95 g
2) Convert the masses to moles:
S ⇒ 50.05 g / 32.066 g/mol = 1.5608 mol
O ⇒ 49.95 g / 16.00 g/mol = 3.1212 mol
3) Divide by the lowest, seeking the smallest whole-number ratio:
S ⇒ 1.5608 / 1.5608 = 1
O ⇒ 3.1212 / 1.5608 = 2
4) Write the empirical formula:
SO2
5) Compute the "empirical formula weight:"
32 + 16 + 16 = 64
6) Divide the molecule weight by the "EFW:"
64.07 / 64 = 1
7) Use the scaling factor computed just above to determine the molecular formula:
SO2 times 1 gives SO2 for the molecular formula
__________________________________________________
Example 2:
A compound is found to contain 64.80 % carbon, 13.62 % hydrogen, and 21.58 % oxygen by weight. What is the empirical formula for this compound? The molecular weight for this compound is 74.14 g/mol. What is its molecular formula?
Solution:
1) Assume 100 g of the compound is present. This changes the percents to grams:
C ⇒ 64.80 g
H ⇒ 13.62 g
O ⇒ 21.58 g
2) Convert the masses to moles:
C ⇒ 64.80 g / 12 = 5.4
H ⇒ 13.62 g / 1 = 13.62
O ⇒ 21.58 g / 16 = 1.349
3) Divide by the lowest, seeking the smallest whole-number ratio:
C ⇒ 5.4 / 1.349 = 4
H ⇒ 13.62 / 1.349 = 10
O ⇒ 1.349 / 1.349 = 1
4) Write the empirical formula:
C4H10O
5) Determine the molecular formula:
"EFW" ⇒ 48+10+16 = 74 --> 74.14 / 74 = 1
molecular formula = C4H10O
Chemists don't deal with the masses of atoms and molecules measured in grams because they would be dealing with extremely small numbers. Instead, the masses of atoms and molecules are compared with that of the isotope carbon-12. 12C is assigned a relative atomic mass of 12. As an example, consider an atom of magnesium. Two atoms of 12C are required to be equal in mass to one atom of Mg therefore the relative atomic mass of Mg is 24.
U1.2.4 The empirical formula and molecular formula of a compound give the simplest ratio and the actual number of atoms present in a molecule respectively.
(Application and Skill) AS1.2.3 Interconversion of the percentage composition by mass and the empirical formula.
AS1.2.4 Determination of the molecular formula of a compound from its empirical formula and molar mass.
Example 1:
A compound is found to contain 50.05 % sulfur and 49.95 % oxygen by weight. What is the empirical formula for this compound? The molecular weight for this compound is 64.07 g/mol. What is its molecular formula?
Solution:
1) Assume 100 g of the compound is present. This changes the percents to grams:
S ⇒ 50.05 g
O ⇒ 49.95 g
2) Convert the masses to moles:
S ⇒ 50.05 g / 32.066 g/mol = 1.5608 mol
O ⇒ 49.95 g / 16.00 g/mol = 3.1212 mol
3) Divide by the lowest, seeking the smallest whole-number ratio:
S ⇒ 1.5608 / 1.5608 = 1
O ⇒ 3.1212 / 1.5608 = 2
4) Write the empirical formula:
SO2
5) Compute the "empirical formula weight:"
32 + 16 + 16 = 64
6) Divide the molecule weight by the "EFW:"
64.07 / 64 = 1
7) Use the scaling factor computed just above to determine the molecular formula:
SO2 times 1 gives SO2 for the molecular formula
__________________________________________________
Example 2:
A compound is found to contain 64.80 % carbon, 13.62 % hydrogen, and 21.58 % oxygen by weight. What is the empirical formula for this compound? The molecular weight for this compound is 74.14 g/mol. What is its molecular formula?
Solution:
1) Assume 100 g of the compound is present. This changes the percents to grams:
C ⇒ 64.80 g
H ⇒ 13.62 g
O ⇒ 21.58 g
2) Convert the masses to moles:
C ⇒ 64.80 g / 12 = 5.4
H ⇒ 13.62 g / 1 = 13.62
O ⇒ 21.58 g / 16 = 1.349
3) Divide by the lowest, seeking the smallest whole-number ratio:
C ⇒ 5.4 / 1.349 = 4
H ⇒ 13.62 / 1.349 = 10
O ⇒ 1.349 / 1.349 = 1
4) Write the empirical formula:
C4H10O
5) Determine the molecular formula:
"EFW" ⇒ 48+10+16 = 74 --> 74.14 / 74 = 1
molecular formula = C4H10O

APPLICATION AND SKILL:
AS1.2.1 Calculation of the molar masses of atoms, ions, molecules and formula units.
AS1.2.2 Solution of problems involving the relationships between the number of particles, the amount of substance in moles and the mass in grams.
Calculate the number of ATOMS in:
18.0g of carbon
1 mole of carbon = 12.01g
(18 ÷ 12.01) x (6.02 x 10^23)
= 9.02 x 10^23 particles
_______________________________________
Calculate the number of MOLECULES in:
3.28g of sulphur dioxide
(3.28 ÷ 64.06) x (6.02 x 10^23)
= 3.08 x 10^22 molecules
_______________________________________
Calculate the number of IONS present in:
18.5g of calcium chloride, CaCl2
(18.5 ÷ 110.98) x 3 x (6.02 x 10^23)
= 3.01 x 10^23 ions
We multiply this equation by 3 because there are THREE ions present in the formula - Ca and 2 x Cl
AS1.2.1 Calculation of the molar masses of atoms, ions, molecules and formula units.
AS1.2.2 Solution of problems involving the relationships between the number of particles, the amount of substance in moles and the mass in grams.
Calculate the number of ATOMS in:
18.0g of carbon
1 mole of carbon = 12.01g
(18 ÷ 12.01) x (6.02 x 10^23)
= 9.02 x 10^23 particles
_______________________________________
Calculate the number of MOLECULES in:
3.28g of sulphur dioxide
(3.28 ÷ 64.06) x (6.02 x 10^23)
= 3.08 x 10^22 molecules
_______________________________________
Calculate the number of IONS present in:
18.5g of calcium chloride, CaCl2
(18.5 ÷ 110.98) x 3 x (6.02 x 10^23)
= 3.01 x 10^23 ions
We multiply this equation by 3 because there are THREE ions present in the formula - Ca and 2 x Cl
AS1.2.5 Obtaining and using experimental data for deriving empirical formulas from reactions involving mass changes.
Experiments to be done in class.
Experiments to be done in class.
Essential idea: Mole ratios in chemical equations can be used to calculate reacting ratios by mass and gas volume.

1.3 Reacting masses and volumes
U1.3.1 Reactants can be either limiting or excess.
Example Problem #2
Methane, CH4, burns in oxygen to give carbon dioxide and water according to the following equation:
CH4 + 2O2 ------> CO2 + 2H2O
In one experiment, a mixture of 0.250 mol of methane was burned in 1.25 mol of oxygen in a sealed steel vessel. Find the limiting reactant, if any, and calculate the theoretical yield, (in moles) of water.
Solution:
In any limiting reactant question, the decision can be stated in two ways. Do it once to get an answer, then do it again the second way to get a confirmation.
According to the equation: 1 mol CH4 = 2 mol O2
If we use up all the methane then:
1 mol CH4 = 2 mol O2
0.25 mol x x = 0.50 mol of O2 would be needed.
We have 1.25 mol of O2 on hand. Therefore we have 0.75 mol of O2 in excess of what we need.
If the oxygen in is excess, then the methane is the limiting reactant.
Confirmation: If we use up all the oxygen then 1 mol CH4 = 2 mol O2
x 1.25 mol x = 0.625 mol of methane.
We don't have 0.625 moles of methane. We have only 0.25 moles. Therefore the methane will be used up before all the oxygen is. Again the methane is the limiting reactant.
OR...There is an even simpler way to do this:
Step 1. Begin by writing your balanced chemical equation. This will serve as a mathematical representation of your eventual reaction.
Step 2: Identify your reactants. These are generally on the left side of the arrow in the equation. These are also referred to as "starting materials."
Step 3: Calculate the amount (in moles) of each reactant. To do this, divide each reactant's mass by its molar mass.
Step 4: If you have two reactants, compare the ratio of your reactants' moles to the ratio of their coefficients. Respectively, these ratios are called the "actual ratio" and the "stoichiometric ratio."
U1.3.1 Reactants can be either limiting or excess.
Example Problem #2
Methane, CH4, burns in oxygen to give carbon dioxide and water according to the following equation:
CH4 + 2O2 ------> CO2 + 2H2O
In one experiment, a mixture of 0.250 mol of methane was burned in 1.25 mol of oxygen in a sealed steel vessel. Find the limiting reactant, if any, and calculate the theoretical yield, (in moles) of water.
Solution:
In any limiting reactant question, the decision can be stated in two ways. Do it once to get an answer, then do it again the second way to get a confirmation.
According to the equation: 1 mol CH4 = 2 mol O2
If we use up all the methane then:
1 mol CH4 = 2 mol O2
0.25 mol x x = 0.50 mol of O2 would be needed.
We have 1.25 mol of O2 on hand. Therefore we have 0.75 mol of O2 in excess of what we need.
If the oxygen in is excess, then the methane is the limiting reactant.
Confirmation: If we use up all the oxygen then 1 mol CH4 = 2 mol O2
x 1.25 mol x = 0.625 mol of methane.
We don't have 0.625 moles of methane. We have only 0.25 moles. Therefore the methane will be used up before all the oxygen is. Again the methane is the limiting reactant.
OR...There is an even simpler way to do this:
Step 1. Begin by writing your balanced chemical equation. This will serve as a mathematical representation of your eventual reaction.
Step 2: Identify your reactants. These are generally on the left side of the arrow in the equation. These are also referred to as "starting materials."
- If there are numbers next to your reactants, these are called coefficients. These represent the ratios in which your reactants are consumed.
- The limiting reactant is the starting material which will run out first. In reactions with multiple starting materials, unless every starting material is consumed in 1:1 ratios with every other and is present in the same molar quantity, one reactant will be consumed first, at which point the reaction will stop.
Step 3: Calculate the amount (in moles) of each reactant. To do this, divide each reactant's mass by its molar mass.
- To calculate a molecule's molar mass, add the atomic masses of each atom in the compound. An atom's atomic mass is the decimal number under its chemical symbol on the periodic table. Remember that any subscripted numbers in a molecule mean that those atoms occur more than once.
Step 4: If you have two reactants, compare the ratio of your reactants' moles to the ratio of their coefficients. Respectively, these ratios are called the "actual ratio" and the "stoichiometric ratio."
- If the actual ratio is greater than the stoichiometric ratio, the reactant in the numerator is "in excess," meaning the other reactant is the limiting reactant.
- If the actual ratio is smaller than the stoichiometric ratio, the reactant in the numerator is the limiting reactant.
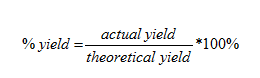
U1.3.2 The experimental yield can be different from the theoretical yield.
Reasons that experimental yields may be different from theoretical yields include:
Example problem #1
16g of methane burns completely in oxygen. The water produced is condensed and weighed. 30g of water is collected.
1. Balanced equation:
CH4 + 2O2 --> 2H2O + CO2
2. Calculate theoretical yield of water:
CH4 Mr = 16.05
H2O Mr = 18.02
CH4 mol = 16/16.05 = 0.99 mol
H2O mol (using mol ratio) = 1.99 mol
n x Mr = mass of H2O = 1.99 x 18.02 = 35.9g
3. Calculate percentage yield of water:
(30/35.9) x 100 = 83.6%
U1.3.3 Avogadro’s law enables the mole ratio of reacting gases to be determined from volumes of the gases.
Reasons that experimental yields may be different from theoretical yields include:
- loss of reactants during transfers
- incomplete reactions (includig reversible reactions)
- side reactions
- impurities in starting materials
Example problem #1
16g of methane burns completely in oxygen. The water produced is condensed and weighed. 30g of water is collected.
1. Balanced equation:
CH4 + 2O2 --> 2H2O + CO2
2. Calculate theoretical yield of water:
CH4 Mr = 16.05
H2O Mr = 18.02
CH4 mol = 16/16.05 = 0.99 mol
H2O mol (using mol ratio) = 1.99 mol
n x Mr = mass of H2O = 1.99 x 18.02 = 35.9g
3. Calculate percentage yield of water:
(30/35.9) x 100 = 83.6%
U1.3.3 Avogadro’s law enables the mole ratio of reacting gases to be determined from volumes of the gases.
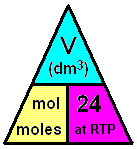
This formula is applicable for any gas at room temperature and pressure (RTP) (Avogadro's Law)
Room temperature = 298 Kelvin (K)
Room pressure = 1 atmosphere (atm)
NOTE: 1 mole of any gas occupies 22.4 dm3 at STP (0 degrees, 1 atm) or equal volumes of any gas must contain the same number of particles under the same conditions of temperature and pressure.
E.g.
How many moles are there in 12dm3 of carbon dioxide?
n = v/24 = 12/24 = 0.5 mol
NOTE: It doesn't matter what the gas is, one mole of any gas occupies 24dm3 at RTP.
Example problem #1
What volume of carbon dioxide reacts with 36g of magnesium at rtp?
2Mg(s) + CO2(g) --> 2MgO(s) + C(s)
Mg mol = 36/24.31 = 1.48
Mol ratio = 2:1
CO2 mol = 0.740
Volume = n x 24 = 0.740 x 24 = 17.8dm3
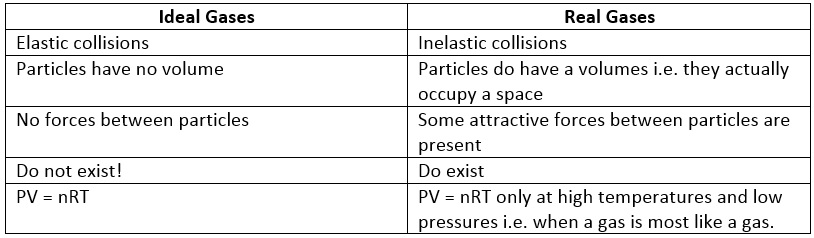
U1.3.4 The molar volume of an ideal gas is a constant at specified temperature and pressure.
Kinetic Theory assumptions about ideal gases:
There is no such thing as an ideal gas, of course, but many gases behave approximately as if they were ideal at ordinary working temperatures and pressures. Real gases are dealt with in more detail on another page.
The assumptions are:
The Ideal Gas Equation
PV = nRT
R = gas constant
P = pressure (measured in either pascal, atm or Nm^-2 --> 1.01 x 10^5 Nm^-2/Pa)
V = volume
n = number of moles
R = gas constant (a value for R will be given you if you need it, or you can look it up in a data source. The SI value for R is 8.31441 J K-1 mol-1)
T = temperature
A gas which obeys the ideal gas equation is called an 'Ideal Gas'. In practice, real gases obey this equation quite closely only at low pressure and high temperature (i.e. when the gas is most like a gas and least like a liquid).
NOTE: The actual definition of temperature is:
A measure of the AVERAGE kinetic energy of particles in a sample.
Limitations of the Ideal Model
For most applications, the ideal gas approximation is reasonably accurate; the ideal gas model tends to fail at lower temperatures and higher pressures, however, when intermolecular forces and the excluded volume of gas particles become significant. The model also fails for most heavy gases (including many refrigerants) and for gases with strong intermolecular forces (such as water vapor). At a certain point of combined low temperature and high pressure, real gases undergo a phase transition from the gaseous state into the liquid or solid state. The ideal gas model, however, does not describe or allow for phase transitions; these must be modeled by more complex equations of state.
Room temperature = 298 Kelvin (K)
Room pressure = 1 atmosphere (atm)
NOTE: 1 mole of any gas occupies 22.4 dm3 at STP (0 degrees, 1 atm) or equal volumes of any gas must contain the same number of particles under the same conditions of temperature and pressure.
E.g.
How many moles are there in 12dm3 of carbon dioxide?
n = v/24 = 12/24 = 0.5 mol
NOTE: It doesn't matter what the gas is, one mole of any gas occupies 24dm3 at RTP.
Example problem #1
What volume of carbon dioxide reacts with 36g of magnesium at rtp?
2Mg(s) + CO2(g) --> 2MgO(s) + C(s)
Mg mol = 36/24.31 = 1.48
Mol ratio = 2:1
CO2 mol = 0.740
Volume = n x 24 = 0.740 x 24 = 17.8dm3
U1.3.4 The molar volume of an ideal gas is a constant at specified temperature and pressure.
Kinetic Theory assumptions about ideal gases:
There is no such thing as an ideal gas, of course, but many gases behave approximately as if they were ideal at ordinary working temperatures and pressures. Real gases are dealt with in more detail on another page.
The assumptions are:
- Gases consist of molecules in random motion; these particles undergo frequent collisions with each other and with the walls of their container
- Collisions between gas particles are elastic (no loss of kinetic energy)
- The volume occupied by individual gas particles is negligible
- The (attractive) forces between particles are negligible
- The average kinetic energy of gas particles is directly proportional to the absolute temperature
- At a given temperature, all gases have the same average kinetic energy.
- There are no (or entirely negligible) intermolecular forces between the gas molecules
- The volume occupied by the molecules themselves is entirely negligible relative to the volume of the container
The Ideal Gas Equation
PV = nRT
R = gas constant
P = pressure (measured in either pascal, atm or Nm^-2 --> 1.01 x 10^5 Nm^-2/Pa)
V = volume
n = number of moles
R = gas constant (a value for R will be given you if you need it, or you can look it up in a data source. The SI value for R is 8.31441 J K-1 mol-1)
T = temperature
A gas which obeys the ideal gas equation is called an 'Ideal Gas'. In practice, real gases obey this equation quite closely only at low pressure and high temperature (i.e. when the gas is most like a gas and least like a liquid).
NOTE: The actual definition of temperature is:
A measure of the AVERAGE kinetic energy of particles in a sample.
Limitations of the Ideal Model
For most applications, the ideal gas approximation is reasonably accurate; the ideal gas model tends to fail at lower temperatures and higher pressures, however, when intermolecular forces and the excluded volume of gas particles become significant. The model also fails for most heavy gases (including many refrigerants) and for gases with strong intermolecular forces (such as water vapor). At a certain point of combined low temperature and high pressure, real gases undergo a phase transition from the gaseous state into the liquid or solid state. The ideal gas model, however, does not describe or allow for phase transitions; these must be modeled by more complex equations of state.
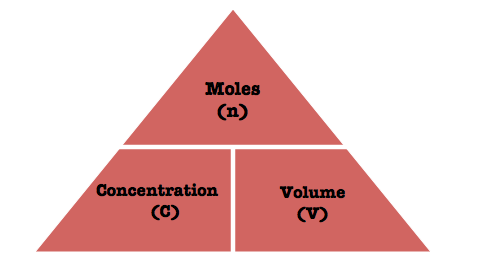
U1.3.5 The molar concentration of a solution is determined by the amount of solute and the volume of solution.
A solution contains a dissolved solute in a certain amount of solvent.
The concentration of a solution tells us how many moles of the solute are dissolved in one litre (1dm3) of the solvent.
The units for concentration are mol dm-3 (mol per dm3) and this is often shortened to M.
Concentration can also be measured in grams per litre or g dm-3.
In this triangle, volume MUST be in dm3 and concentration MUST be in mol dm-3.
U1.3.6 A standard solution is one of known concentration.
Definition of standard solution: In analytical chemistry, a standard solution is a solution containing a precisely known concentration of an element or a substance, a known weight of solute is dissolved to make a specific volume. It is prepared using a standard substance, such as a primary standard.
APPLICATION:
A1.3.1 Solution of problems relating to reacting quantities, limiting and excess reactants, theoretical, experimental and percentage yields.
A1.3.2 Calculation of reacting volumes of gases using Avogadro’s law.
See above points.
A1.3.3 Solution of problems and analysis of graphs involving the relationship between temperature, pressure and volume for a fixed mass of an ideal gas.
SEE TEACHER.
A1.3.4 Solution of problems relating to the ideal gas equation.
A1.3.6 Obtaining and using experimental values to calculate the molar mass of a gas from the ideal gas equation.

A1.3.5 Explanation of the deviation of real gases from ideal behaviour at low temperature and high pressure.
A1.3.7 Solution
of problems involving molar concentration, amount of solute and volume of
solution.
A1.3.8 Use of the experimental method of titration to calculate the concentration of a solution by reference to a standard solution.
A1.3.8 Use of the experimental method of titration to calculate the concentration of a solution by reference to a standard solution.