Essential idea: The mass of an atom is concentrated in its minute, positively charged nucleus.
2.1 The nuclear atom
UNDERSTANDINGS:
U2.1.1 Atoms contain a positively charged dense nucleus composed of protons and neutrons (nucleons).
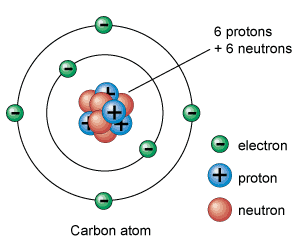
Atomic nuclei are composed of two types of particles, protons and neutrons, which are collectively known as nucleons. A proton is simply the nucleus of an ordinary hydrogen atom, the lightest atom, and has a unit positive charge. A neutron is an uncharged particle of about the same mass as the proton. The number of protons in a given nucleus is the atomic number of that nucleus and determines which chemical element the nucleus will constitute when surrounded by electrons.
UNDERSTANDINGS:
U2.1.1 Atoms contain a positively charged dense nucleus composed of protons and neutrons (nucleons).
Atomic nuclei are composed of two types of particles, protons and neutrons, which are collectively known as nucleons. A proton is simply the nucleus of an ordinary hydrogen atom, the lightest atom, and has a unit positive charge. A neutron is an uncharged particle of about the same mass as the proton. The number of protons in a given nucleus is the atomic number of that nucleus and determines which chemical element the nucleus will constitute when surrounded by electrons.
U2.1.2 Negatively charged electrons occupy the space outside the
nucleus.
The 3rd subatomic particle resides outside of the nucleus in the electron cloud. An electron is the subatomic particle with a negative charge and relatively no mass.
In an atom:
• The protons = the electrons
• If 20 protons are present in an atom then 20 electrons are there to balance the overall charge of the atom—atoms are neutral
• The neutrons have no charge; therefore they do not have to equal the number of protons or electrons.
U2.1.3 The mass spectrometer is used to determine the relative atomic mass of an element from its isotopic composition.
The 3rd subatomic particle resides outside of the nucleus in the electron cloud. An electron is the subatomic particle with a negative charge and relatively no mass.
In an atom:
• The protons = the electrons
• If 20 protons are present in an atom then 20 electrons are there to balance the overall charge of the atom—atoms are neutral
• The neutrons have no charge; therefore they do not have to equal the number of protons or electrons.
U2.1.3 The mass spectrometer is used to determine the relative atomic mass of an element from its isotopic composition.