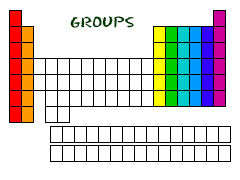periodicity
3.1 Periodic table
UNDERSTANDINGS:
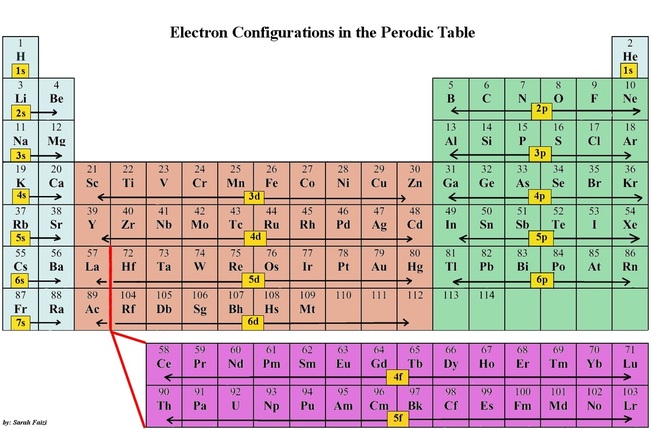
U3.1.1 The periodic table is arranged into four blocks associated with the four sub-levels—s, p, d, and f.
Occupation of Orbitals:
Electrons fill orbitals in a way to minimize the energy of the atom. Therefore, the electrons in an atom fill the principal energy levels in order of increasing energy (the electrons are getting farther from the nucleus). The order of levels filled looks like this:
1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, and 7p
U3.1.2 The periodic table consists of groups (vertical columns) and periods (horizontal rows).
U3.1.3 The period number (n) is the outer energy level that is occupied by electrons.
U3.1.4 The number of the principal energy level and the number of the valence electrons in an atom can be deduced from its position on the periodic table.
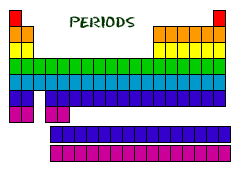
Periods
Rows of elements are called periods. All elements in the same row have their highest energy electron in the same principal energy level. The number of elements in a period increases as you move down the periodic table because there are more sublevels per level as the energy level of the atom increases.
Groups
Columns of elements help define element groups. Elements within a group shareseveral common properties. Groups are elements have the same outer electron arrangement. The outer electrons are called valence electrons. Because they have the same number of valence electrons, elements in a group share similar chemical properties.
UNDERSTANDINGS:
U3.1.1 The periodic table is arranged into four blocks associated with the four sub-levels—s, p, d, and f.
Occupation of Orbitals:
Electrons fill orbitals in a way to minimize the energy of the atom. Therefore, the electrons in an atom fill the principal energy levels in order of increasing energy (the electrons are getting farther from the nucleus). The order of levels filled looks like this:
1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, and 7p
U3.1.2 The periodic table consists of groups (vertical columns) and periods (horizontal rows).
U3.1.3 The period number (n) is the outer energy level that is occupied by electrons.
U3.1.4 The number of the principal energy level and the number of the valence electrons in an atom can be deduced from its position on the periodic table.
Periods
Rows of elements are called periods. All elements in the same row have their highest energy electron in the same principal energy level. The number of elements in a period increases as you move down the periodic table because there are more sublevels per level as the energy level of the atom increases.
Groups
Columns of elements help define element groups. Elements within a group shareseveral common properties. Groups are elements have the same outer electron arrangement. The outer electrons are called valence electrons. Because they have the same number of valence electrons, elements in a group share similar chemical properties.