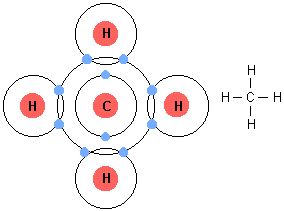
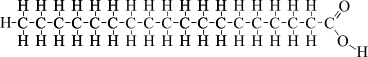
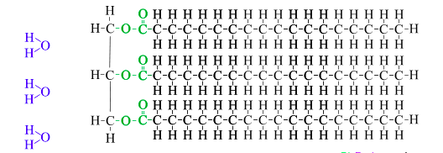
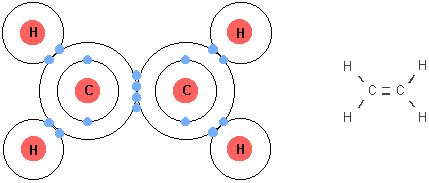
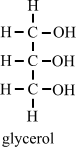Essential idea: Living organisms control their composition by a complex web of chemical reactions.
2.1 Molecules to metabolism
UNDERSTANDINGS:
U2.1.1 Molecular biology explains living processes in terms of the chemical substances involved.
The most frequently occurring chemical elements in living things are:
A variety of other elements are needed by living organisms including:
There are many molecules important to living organisms including water, carbohydrates, lipids, proteins and nucleic acids. Proteins are one of the most varied macromolecules, performing many cellular functions, including catalyzing metabolic reactions (enzymes). The relationship between genes and proteins is also bery significant and will be discussed later in this topic. Molecular biologists break down biochemical processes into their component parts (reductionism). When they look at the sum of all these reactions as a whole, they can study the emergent properties of that system.
U2.1.2 Carbon atoms can form four covalent bonds allowing a diversity of stable compounds to exist.
UNDERSTANDINGS:
U2.1.1 Molecular biology explains living processes in terms of the chemical substances involved.
The most frequently occurring chemical elements in living things are:
- carbon
- hydrogen
- oxygen
- nitrogen
A variety of other elements are needed by living organisms including:
- phosphorus
- sulfur
- iron
- calcium
- sodium
There are many molecules important to living organisms including water, carbohydrates, lipids, proteins and nucleic acids. Proteins are one of the most varied macromolecules, performing many cellular functions, including catalyzing metabolic reactions (enzymes). The relationship between genes and proteins is also bery significant and will be discussed later in this topic. Molecular biologists break down biochemical processes into their component parts (reductionism). When they look at the sum of all these reactions as a whole, they can study the emergent properties of that system.
U2.1.2 Carbon atoms can form four covalent bonds allowing a diversity of stable compounds to exist.
- Carbon has a few unique bonding properties - the most important of which is its ability to form long chains of carbon.
- The reason carbon can do this is that carbon-carbon bonds are extremely strong. This allows carbon to make up many of the basic building blocks of life (fats, sugars, etc).
- Since carbon-carbon bonds are strong and stable, carbon can form an almost infinite number of compounds
- In fact, there are more known carbon-containing compounds than all the compounds of the other chemical elements combined except those of hydrogen (because almost all organic compounds contain hydrogen too).
- Carbon can also form rings e.g. glucose
- The simplest form of an organic molecule is the hydrocarbon—a large family of organic molecules that are composed of hydrogen atoms bonded to a chain of carbon atoms. e.g. methane
- All bonding in hydrocarbons is covalent
- Covalent Bonds are chemical bonds formed by the sharing of a pair of electrons between atoms. The nuclei of two different atoms are attracting the same electrons.
- Carbon can form single, double and triple bonds.
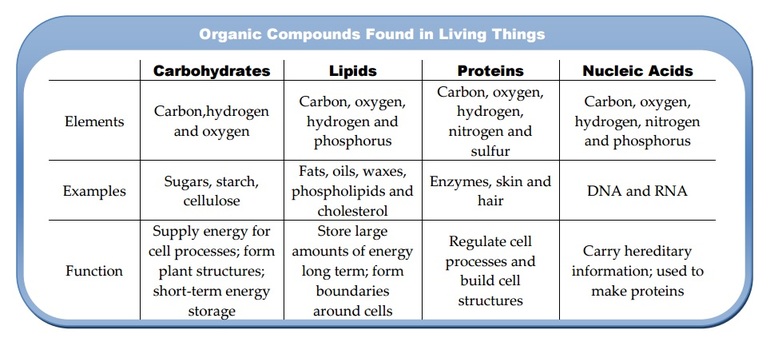
U2.1.3 Life
is based on carbon compounds including carbohydrates, lipids, proteins and
nucleic acids.
Carbohydrates:
Carbohydrates are organic molecules that supply energy for cell processes. Sugars and starches are carbohydrates that cells use for energy. Some carbohydrates also are important parts of cell structures. For example, a carbohydrate called cellulose is an important part of plant cells.
Lipids:
Another type of organic compound found in living things is a lipid. Lipids do not mix with water. Lipids such as fats and oils store and release even larger amounts of energy than carbohydrates do. One type of lipid, the phospholipid, is a major part of cell membranes.
Proteins:
Organic compounds called proteins have many important functions in living organisms. They are made of smaller molecules called amino acids. Proteins are the building blocks of many structures in organisms. Your muscles contain large amount of protein. Proteins are scattered throughout cell membranes. Certain proteins called enzymes regulate nearly all chemical reactions in cells.
Nucleic Acids:
Large organic molecules that store important coded information in cells are called nucleic acids. One nucleic acid, deoxyribonucleic acid, or DNA – genetic material – is found in all cells. It carries information that directs each cell’s activities. Another nucleic acid, ribonucleic acid, or RNA is needed to make enzymes and other proteins.
Carbohydrates:
Carbohydrates are organic molecules that supply energy for cell processes. Sugars and starches are carbohydrates that cells use for energy. Some carbohydrates also are important parts of cell structures. For example, a carbohydrate called cellulose is an important part of plant cells.
Lipids:
Another type of organic compound found in living things is a lipid. Lipids do not mix with water. Lipids such as fats and oils store and release even larger amounts of energy than carbohydrates do. One type of lipid, the phospholipid, is a major part of cell membranes.
Proteins:
Organic compounds called proteins have many important functions in living organisms. They are made of smaller molecules called amino acids. Proteins are the building blocks of many structures in organisms. Your muscles contain large amount of protein. Proteins are scattered throughout cell membranes. Certain proteins called enzymes regulate nearly all chemical reactions in cells.
Nucleic Acids:
Large organic molecules that store important coded information in cells are called nucleic acids. One nucleic acid, deoxyribonucleic acid, or DNA – genetic material – is found in all cells. It carries information that directs each cell’s activities. Another nucleic acid, ribonucleic acid, or RNA is needed to make enzymes and other proteins.
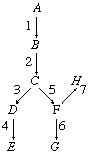
U2.1.4 Metabolism is the web of all the enzyme-catalysed reactions in a cell or organism.
The role of enzymes in metabolic pathways can be illustrated diagrammatically. The chemical compound represented by A (see diagram) is converted to product E in a series of enzyme-catalyzed steps, in which intermediate compounds represented byB, C, and D are formed in succession. They act as substrates for enzymes represented by 2, 3, and 4. Compound A may also be converted by another series of steps, some of which are the same as those in the pathway for the formation of E, to products represented by G and H.
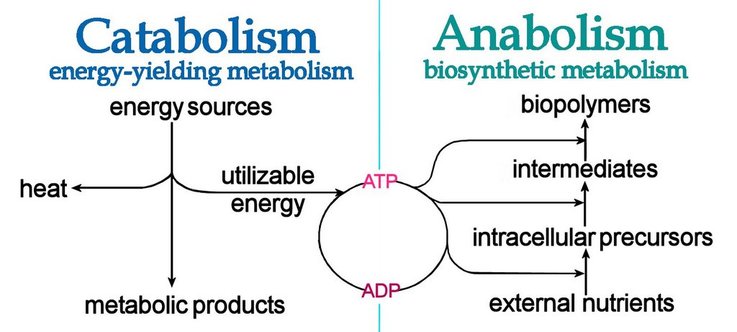
U2.1.5 Anabolism is the synthesis of complex molecules from simpler molecules including the formation of macromolecules from monomers by condensation reactions.
U2.1.5 Catabolism is the breakdown of complex molecules into simpler molecules including the hydrolysis of macromolecules into monomers.
- These reactions are catalyzed by enzymes and allow organisms to grow and reproduce, maintain their structures, and respond to their environments.
- Many of these reactions occur in the cytoplasm, but some are extracellular including digestion and the transport of substances into and between different cells.
- The word metabolism can refer to the sum of all chemical reactions that occur in living organisms.
The role of enzymes in metabolic pathways can be illustrated diagrammatically. The chemical compound represented by A (see diagram) is converted to product E in a series of enzyme-catalyzed steps, in which intermediate compounds represented byB, C, and D are formed in succession. They act as substrates for enzymes represented by 2, 3, and 4. Compound A may also be converted by another series of steps, some of which are the same as those in the pathway for the formation of E, to products represented by G and H.
U2.1.5 Anabolism is the synthesis of complex molecules from simpler molecules including the formation of macromolecules from monomers by condensation reactions.
- Metabolism is divided into two components; anabolism (building large molecules from smaller ones) and catabolism (breaking down of large molecules into their component parts).
- Anabolic reactions require energy as you are building large molecules from small ones (takes energy to build things).
- Some anabolic processes are protein synthesis, DNA synthesis and replication, photosynthesis, and building complex carbohydrates, such as cellulose, starch and glycogen.
- If you can’t remember which one is which, think anabolic steroids are used to build muscles in athletes and body builders and catapults are used to break down walls in wars.
U2.1.5 Catabolism is the breakdown of complex molecules into simpler molecules including the hydrolysis of macromolecules into monomers.
- Catabolism are reactions that break down larger molecules into smaller ones or their component parts.
- Catabolic reactions release energy (sometimes captured in the form of ATP).
- Some examples of catabolic reactions are digestion of food, cellular respiration, and break down of carbon compounds by decomposers.
APPLICATION:
A2.1.1 Urea as an example of a compound that is produced by living organisms but can also be artificially synthesized.
Urea is an organic compound found in the urine of animals. It is a nitrogen containing compound and it is produced when there are extra amino acids in the body and the organism needs a way to get rid of the extra nitrogen. Urea is produced in the liver and released into the blood stream. The blood stream carries it to the kidneys where it is filtered out of the blood and passes out of the body in urine.
German chemist Friedrich Wӧhler was trying to synthesize ammonium cyanate (an inorganic salt) in the lab and he ended up making urea instead. This was the first time an organic compound had been synthesized outside of an organism. When Wӧhler published his results it caused quite a stir among vitalists, scientists who believed that organic compounds could only be produced with the help of a “vital principal (living thing). The theory was known as vitalism and it states that the origin and phenomena of life are due to a vital principal.
Friedrich Wӧhler’s finding was significant and while it did not lead to the immediate falsification of the theory of vitalism, it was the first evidence against the theory. It took many more experiments and the synthesis of many more complex organic molecules in the laboratory before the principal was finally abandoned.
SKILL:
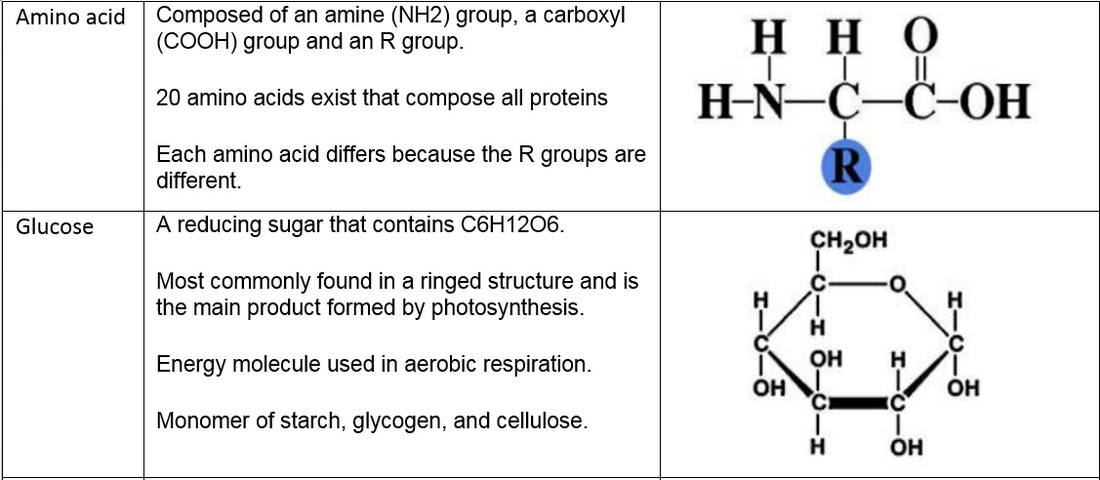
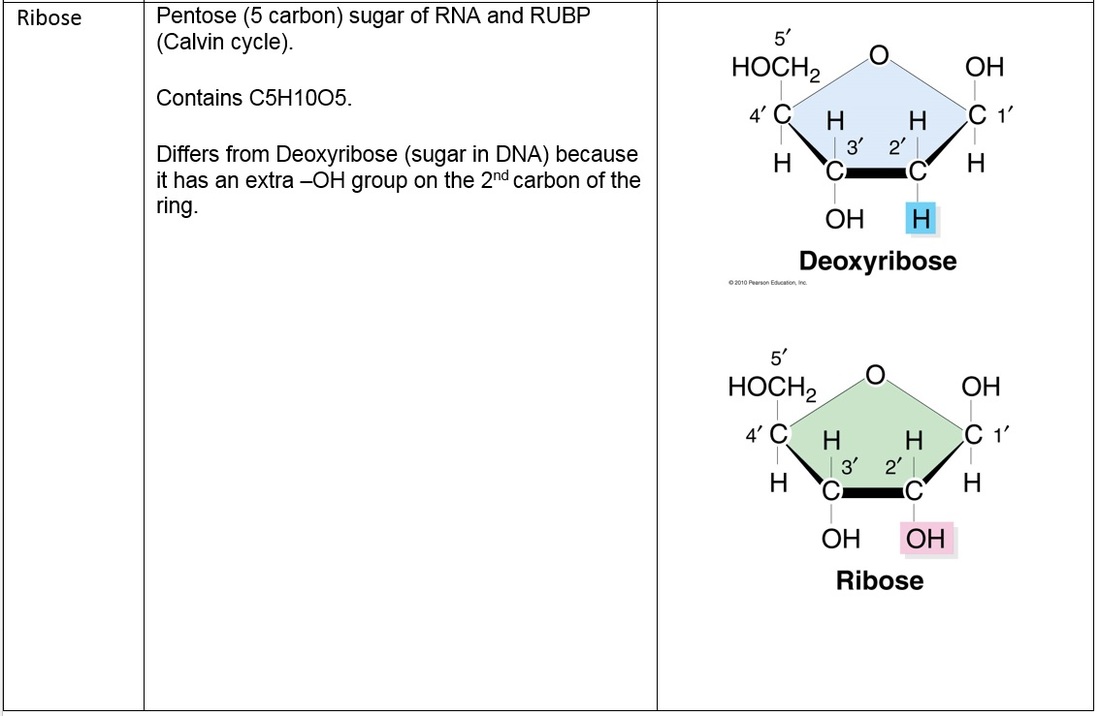
S2.1.1 Drawing molecular diagrams of glucose, ribose, a saturated fatty acid and a generalized amino acid.
S2.1.2 Identification of biochemicals such as sugars, lipids or amino acids from molecular diagrams.
A2.1.1 Urea as an example of a compound that is produced by living organisms but can also be artificially synthesized.
Urea is an organic compound found in the urine of animals. It is a nitrogen containing compound and it is produced when there are extra amino acids in the body and the organism needs a way to get rid of the extra nitrogen. Urea is produced in the liver and released into the blood stream. The blood stream carries it to the kidneys where it is filtered out of the blood and passes out of the body in urine.
German chemist Friedrich Wӧhler was trying to synthesize ammonium cyanate (an inorganic salt) in the lab and he ended up making urea instead. This was the first time an organic compound had been synthesized outside of an organism. When Wӧhler published his results it caused quite a stir among vitalists, scientists who believed that organic compounds could only be produced with the help of a “vital principal (living thing). The theory was known as vitalism and it states that the origin and phenomena of life are due to a vital principal.
Friedrich Wӧhler’s finding was significant and while it did not lead to the immediate falsification of the theory of vitalism, it was the first evidence against the theory. It took many more experiments and the synthesis of many more complex organic molecules in the laboratory before the principal was finally abandoned.
SKILL:
S2.1.1 Drawing molecular diagrams of glucose, ribose, a saturated fatty acid and a generalized amino acid.
S2.1.2 Identification of biochemicals such as sugars, lipids or amino acids from molecular diagrams.

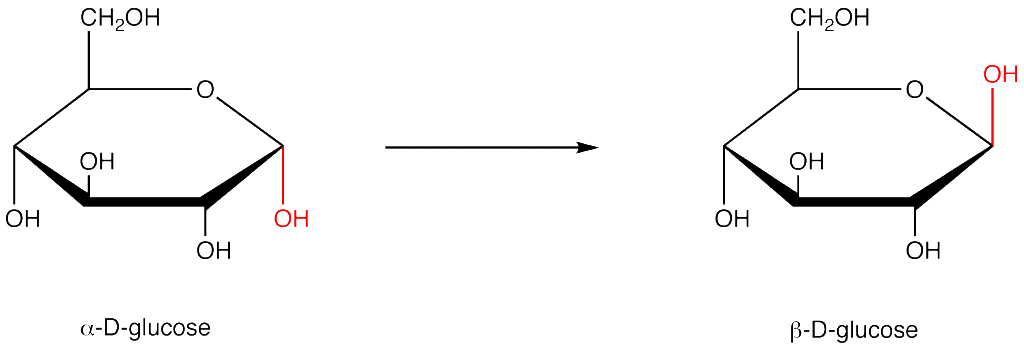
Alpha-D-glucose and Beat-D-glucose are simply isomers of each other.
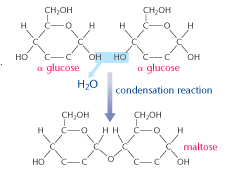
NOTE: When 2 glucose molecules join together in a condensation reaction, they form maltose + H2O (containing GLYCOSIDIC bonds).
NOTE: When 2 glucose molecules join together in a condensation reaction, they form maltose + H2O (containing GLYCOSIDIC bonds).


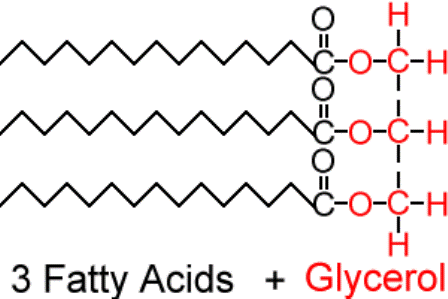
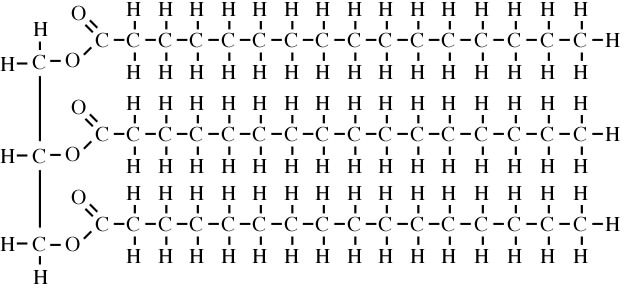
Above is the simple structure of a triglyceride.
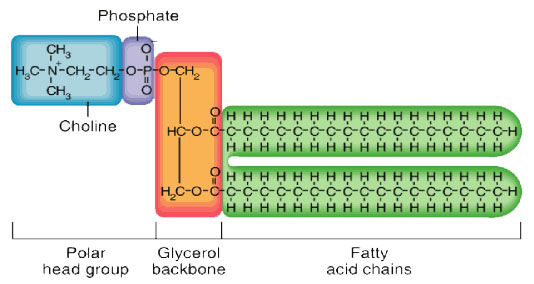
This is the structure of a phospholipid (which is not required to be memorised but should be recognised).
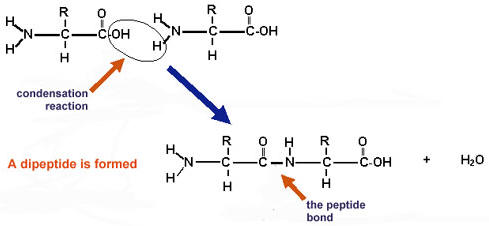
This is the condensation reaction of 2 amino acids joining to form a dipeptide.
NOTE: The process through which polypeptides, polysaccharides or triglycerides are catabolised (broken down) is called HYDROLYSIS.
H2O molecule is absorbed, enzyme breaks polymer bond.
NOTE: The process through which polypeptides, polysaccharides or triglycerides are catabolised (broken down) is called HYDROLYSIS.
H2O molecule is absorbed, enzyme breaks polymer bond.
Essential idea: Water is the medium of life.
2.2 Water
UNDERSTANDINGS:
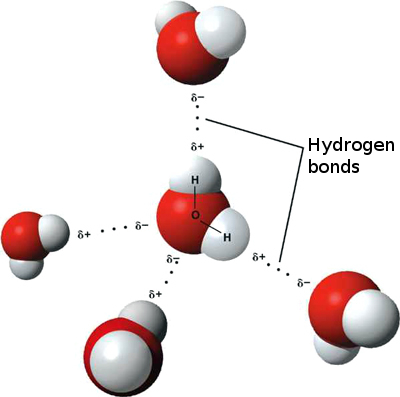
U2.2.1 Water molecules are polar and hydrogen bonds form between them.
UNDERSTANDINGS:
U2.2.1 Water molecules are polar and hydrogen bonds form between them.
- A water molecule consists of an oxygen atom covalently bound to two hydrogen atoms.
- Since O is more electronegative than H, an unequal sharing of electrons occurs.
- This creates a polar covalent bond, with H having a partial positive charge and O having a partial negative charge.
- Water is also bent so the positive charge exists more or less on one side and the negative charge from the O exists on the opposite side.
- The partial +ve charge is attracted to the partial –ve charge creating an intermolecular attraction between the water molecules called a “Hydrogen bond.”
- H-bonds are the strongest of the intermolecular bonding.
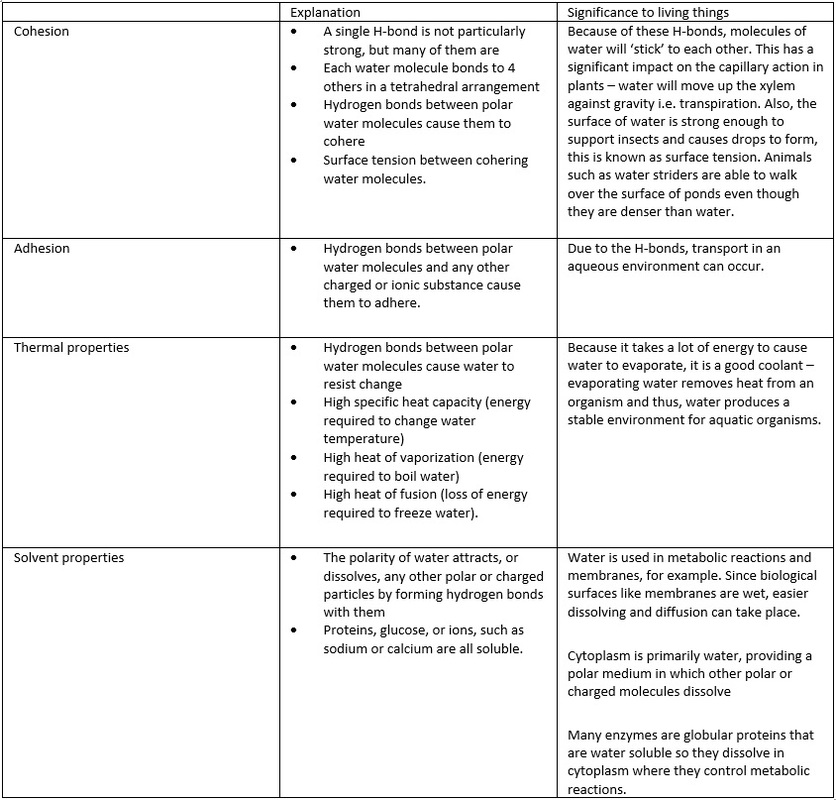
U2.2.2 Hydrogen bonding and dipolarity
explain the cohesive, adhesive, thermal and solvent properties of water.
U2.2.3 Substances can be hydrophilic or hydrophobic.
Essentially hydrophilic means “water loving”. Any substances that dissolves in water including charged ions such as Na+ or polar molecules such as glucose and fructose are hydrophilic. Molecules that are attracted to water likephospholipid heads are also hydrophilic. Hydrophobic molecules are “water fearing” , these are non-polar, insoluble in water or non-charged substances, such as lipids. Try mixing oil and water together - what happens?
Essentially hydrophilic means “water loving”. Any substances that dissolves in water including charged ions such as Na+ or polar molecules such as glucose and fructose are hydrophilic. Molecules that are attracted to water likephospholipid heads are also hydrophilic. Hydrophobic molecules are “water fearing” , these are non-polar, insoluble in water or non-charged substances, such as lipids. Try mixing oil and water together - what happens?
APPLICATION:
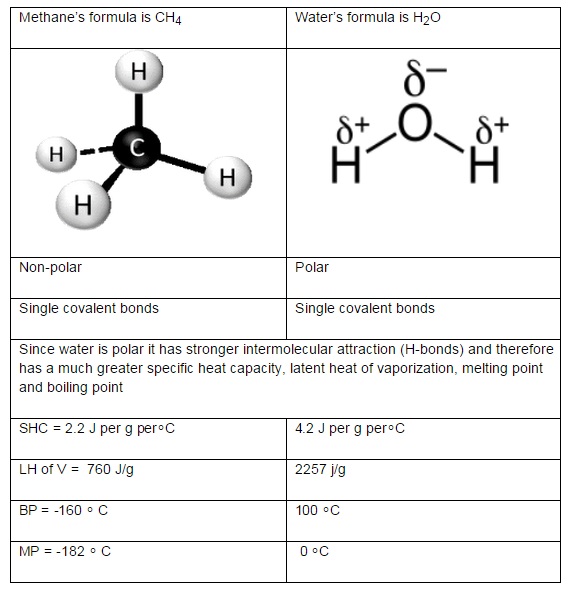
A2.2.1 Comparison of the thermal properties of water with those of methane.
A2.2.2 Use of water as a coolant in sweat.
Water has a high latent heat of vaporization which means it takes a lot of heat to evaporate water from a liquid to a vapour. This is very important as a cooling mechanism for living organisms. As humans sweat, the water droplets absorb heat from the blood flowing under our skin causing the water to evaporate and our blood to cool down. This will in turn cool our whole body down. This cooling is controlled by negative feedback through receptors in the hypothalamusIf the body is overheated, receptors in the hypothalamus sense this and stimulate the sweat glands to secrete sweat. Some reptiles such as crocodiles cool by opening their mouths (gaping). Dogs also pant which causes water to evaporate from their upper respiratory tract.
A2.2.3 Modes of transport of glucose, amino acids, cholesterol, fats, oxygen and sodium chloride in blood in relation to their solubility in water.
Blood transports many different substances to different parts of the body using a variety of methods. Water is critical both as a solvent in which many of the body's solutes dissolve. Due to its polarity water is a great solvent of other polar molecules and ions. This is vital because it allows water to act as a transport medium (blood and cytoplasm) of important molecules in biological organisms.
Glucose:
Glucose is polar, is soluble in water and is therefore transported in the plasma.
Amino acids:
Amino acids have both a negative and a positive charge, but their “R” groups vary, therefore they can be hydrophilic or hydrophobic. They are all soluble enough to be carried in the plasma.
Cholesterol:
Cholesterol, which is mostly hydrophobic because it is a lipid, is also transported inside the lipoprotein complex with the small hydrophilic end facing the phospholipid heads.
Fats:
Fats are nonpolar and therefore insoluble in water. They are transported in a single layer sphere of phospholipids called a lipoprotein complex. The hydrophilic heads face outwards towards the water in the plasma and the tails face inwards towards the fats. Proteins are also embedded in the phospholipid layer.
Oxygen:
Non-compound oxygen, or free oxygen (O2), is oxygen that is not bonded to any other element. Dissolved oxygen is the presence of these free O2 molecules within water.The bonded oxygen molecule in water (H2O) is in a compound and does not count toward dissolved oxygen levels. One can imagine that free oxygen molecules dissolve in water much the way salt or sugar does when it is stirred.
Since blood is mostly water, it dissolves into blood (and not only red blood cells i.e. haemoglobin).
Sodium chloride:
NaCl is an ionic compound that is very soluble in water. Na+ and Cl- dissolve and are carried in the blood plasma.
A2.2.1 Comparison of the thermal properties of water with those of methane.
A2.2.2 Use of water as a coolant in sweat.
Water has a high latent heat of vaporization which means it takes a lot of heat to evaporate water from a liquid to a vapour. This is very important as a cooling mechanism for living organisms. As humans sweat, the water droplets absorb heat from the blood flowing under our skin causing the water to evaporate and our blood to cool down. This will in turn cool our whole body down. This cooling is controlled by negative feedback through receptors in the hypothalamusIf the body is overheated, receptors in the hypothalamus sense this and stimulate the sweat glands to secrete sweat. Some reptiles such as crocodiles cool by opening their mouths (gaping). Dogs also pant which causes water to evaporate from their upper respiratory tract.
A2.2.3 Modes of transport of glucose, amino acids, cholesterol, fats, oxygen and sodium chloride in blood in relation to their solubility in water.
Blood transports many different substances to different parts of the body using a variety of methods. Water is critical both as a solvent in which many of the body's solutes dissolve. Due to its polarity water is a great solvent of other polar molecules and ions. This is vital because it allows water to act as a transport medium (blood and cytoplasm) of important molecules in biological organisms.
Glucose:
Glucose is polar, is soluble in water and is therefore transported in the plasma.
Amino acids:
Amino acids have both a negative and a positive charge, but their “R” groups vary, therefore they can be hydrophilic or hydrophobic. They are all soluble enough to be carried in the plasma.
Cholesterol:
Cholesterol, which is mostly hydrophobic because it is a lipid, is also transported inside the lipoprotein complex with the small hydrophilic end facing the phospholipid heads.
Fats:
Fats are nonpolar and therefore insoluble in water. They are transported in a single layer sphere of phospholipids called a lipoprotein complex. The hydrophilic heads face outwards towards the water in the plasma and the tails face inwards towards the fats. Proteins are also embedded in the phospholipid layer.
Oxygen:
Non-compound oxygen, or free oxygen (O2), is oxygen that is not bonded to any other element. Dissolved oxygen is the presence of these free O2 molecules within water.The bonded oxygen molecule in water (H2O) is in a compound and does not count toward dissolved oxygen levels. One can imagine that free oxygen molecules dissolve in water much the way salt or sugar does when it is stirred.
Since blood is mostly water, it dissolves into blood (and not only red blood cells i.e. haemoglobin).
Sodium chloride:
NaCl is an ionic compound that is very soluble in water. Na+ and Cl- dissolve and are carried in the blood plasma.
Essential idea: Compounds of carbon, hydrogen and oxygen are used to supply and store energy.
2.3 Carbohydrates and lipids
UNDERSTANDINGS:
U2.3.1 Monosaccharide monomers are linked together by condensation reactions to form disaccharides and polysaccharide polymers.
Condensation synthesis:
Hydrolysis:
A condensation reaction is a reaction in which two molecules combine to form a larger molecule, producing a small molecule such as H2O as a byproduct.
Glucose + glucose --> maltose
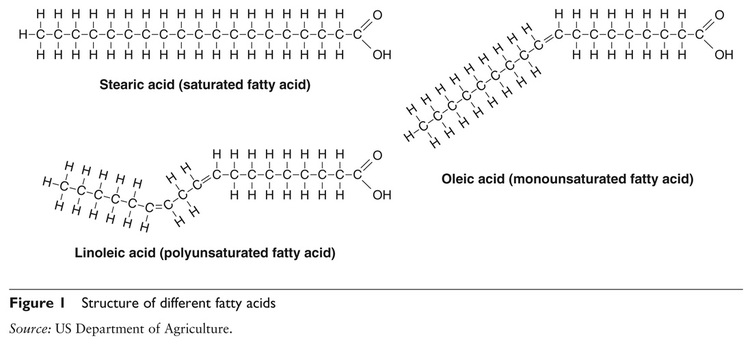
U2.3.2 Fatty acids can be saturated, monounsaturated or polyunsaturated.
Saturated = single bonds between carbon atoms
Monounsaturated = one double bond between 2 carbon atoms
Polyunsaturated = more than one double bond present
UNDERSTANDINGS:
U2.3.1 Monosaccharide monomers are linked together by condensation reactions to form disaccharides and polysaccharide polymers.
Condensation synthesis:
- condensation synthesis reactions link two monosaccharide monomers
- forming one disaccharide molecule and one H2O molecule
- repeated additions of monosaccharides produces a polysaccharide
Hydrolysis:
- a polysaccharides can be broken down into monosaccharides
- H2O molecules used as a sources of -H and a -OH groups
- catalyzed by enzymes
A condensation reaction is a reaction in which two molecules combine to form a larger molecule, producing a small molecule such as H2O as a byproduct.
Glucose + glucose --> maltose
U2.3.2 Fatty acids can be saturated, monounsaturated or polyunsaturated.
Saturated = single bonds between carbon atoms
Monounsaturated = one double bond between 2 carbon atoms
Polyunsaturated = more than one double bond present
U2.3.3 Unsaturated fatty acids can be cis or trans isomers.
While a saturated fatty acid is a straight molecule on the average, the double bond in an unsaturated fatty acid produces a kink in the molecule. This because a double bond cannot rotate. The bend in the carbon chain, however, is much more pronounced in the cis isomer compared to the trans isomer. For this reason, cis fatty acids (and triacylglycerols made from them) do not solidify as readily as trans fatty acids. Due to the larger bend, the cis isomers cannot line up next to one another in as ordered a fashion as the trans isomers.
U2.3.4 Triglycerides are formed by condensation from three fatty acids and one glycerol.
Fats and oils (lipids) are made up from sub-units: fatty acids and glycerol.
While a saturated fatty acid is a straight molecule on the average, the double bond in an unsaturated fatty acid produces a kink in the molecule. This because a double bond cannot rotate. The bend in the carbon chain, however, is much more pronounced in the cis isomer compared to the trans isomer. For this reason, cis fatty acids (and triacylglycerols made from them) do not solidify as readily as trans fatty acids. Due to the larger bend, the cis isomers cannot line up next to one another in as ordered a fashion as the trans isomers.
U2.3.4 Triglycerides are formed by condensation from three fatty acids and one glycerol.
Fats and oils (lipids) are made up from sub-units: fatty acids and glycerol.
APPLICATION:
A2.3.1 Structure and function of cellulose and starch in plants and glycogen in humans.
STARCH:
Starch is a suitable molecule for storing energy as it is composed of two polysaccharides of α glucose - amylose and amylopectin. Amylose is a long, unbranched chain of α glucose. The angles of glycosidic bonds give it a coiled structure - its compact, coiled structure makes it good for storage. Amylopectin is a long, branched chain of α glucose. Its side branches make it good for storage of glucose as the enzymes which breakdown the molecule can reach the glycosidic bonds easily therefore the release of glucose is quick and efficient.
CELLULOSE:
Both cellulose and amylose consist of polypeptide chains of glucose bonded together with glycosidic bonds. Both molecules are insoluble in water. However, Cellulose is composed of β glucose and forms long straight chains, in which every other Glucose monomer flipped over and are very strong, whereas Amylose is composed of α glucose, forms a coiled chain and tends to be found in granules.
Because of their structure, cellulose and amylose have very different functions. Amylose is used as an energy storage in starch, whereas cellulose plays a structural role.
GLYCOGEN:
Glycogen is almost identical to starch but differs in that the chains of (1→4) linked glucoses are shorter, giving it a more highly branched structure. This branching allows for the fast breakdown of the molecule during respiration as it means that there are more ends which enzymes can start the process of hydrolysis from.
A2.3.2 Scientific evidence for health risks of trans fats and saturated fatty acids.
Like saturated or animal fats, trans fats contribute to clogged arteries. Clogged arteries are a sign of heart disease; they increase your risk of both heart attack and stroke. Here's how it works: Trans fats raise low-density lipoprotein (LDL) or "bad" cholesterol levels. This contributes to the buildup of fatty plaque in arteries.
"The science that shows that trans fats increase LDL cholesterol levels is outstanding and very strong. All evidence is pointing in the same direction," Lichtenstein tells WebMD.
In the Nurse's Health Study, women who consumed the greatest amount of trans fats in their diet had a 50% higher risk of heart attack compared to women who consumed the least.
Some researchers suspect that trans fats also increase blood levels of two other artery-clogging compounds -- a fat-protein particle called lipoprotein(a) and blood fats called triglycerides.
Equally worrisome, population studies indicate that trans fats may raise the risk of diabetes. Researchers at the Harvard School of Public Health in Boston suggest that replacing trans fats in the diet with polyunsaturated fats (such as vegetable oils, salmon, etc.) can reduce diabetes risk by as much as 40%.
"Trans fats raise (bad) LDL cholesterol levels slightly less than do saturated fats," says Lichtenstein. "But saturated fats also raise levels of high density lipoprotein (HDL) or "good" cholesterol, and trans fatty acids don't." Trans fats may actually lower HDL.
A2.3.1 Structure and function of cellulose and starch in plants and glycogen in humans.
STARCH:
- Glucose units contain a lot of bonds that can be broken down to release energy during respiration to create ATP. The breakdown occurs in a series of steps which are driven by shape-specific enzymes. In plants and animals, only α glucose can be broken down in respiration as only the enzymes which fit its shape are present.
- α Glucose can form long chains with thousands of subunits called an amylose molecule. Glucose units are bonded together by condensation reactions forming (1→4) glycosidic bonds. Amylose molecules tend to form coiled springs due to the way in which the the glucose units bond, making it quite compact. Large molecules such as amylose differ from glucose in that they are not water soluble.
- Iodine molecules can become trapped within the 'coils' of the amylose chain, which causes iodine (in Potassium Iodide solution) to change colour from yellow-brown to blue-black. Hence the result of the test for starch.
- Starch consists of a mixture of amylose and a branched carbohydrate chain called amylopectin. The branches are formed when one end of a chain joins with a glucose in another, forming a (1→4) glycosidic bond.
Starch is a suitable molecule for storing energy as it is composed of two polysaccharides of α glucose - amylose and amylopectin. Amylose is a long, unbranched chain of α glucose. The angles of glycosidic bonds give it a coiled structure - its compact, coiled structure makes it good for storage. Amylopectin is a long, branched chain of α glucose. Its side branches make it good for storage of glucose as the enzymes which breakdown the molecule can reach the glycosidic bonds easily therefore the release of glucose is quick and efficient.
CELLULOSE:
Both cellulose and amylose consist of polypeptide chains of glucose bonded together with glycosidic bonds. Both molecules are insoluble in water. However, Cellulose is composed of β glucose and forms long straight chains, in which every other Glucose monomer flipped over and are very strong, whereas Amylose is composed of α glucose, forms a coiled chain and tends to be found in granules.
Because of their structure, cellulose and amylose have very different functions. Amylose is used as an energy storage in starch, whereas cellulose plays a structural role.
GLYCOGEN:
Glycogen is almost identical to starch but differs in that the chains of (1→4) linked glucoses are shorter, giving it a more highly branched structure. This branching allows for the fast breakdown of the molecule during respiration as it means that there are more ends which enzymes can start the process of hydrolysis from.
A2.3.2 Scientific evidence for health risks of trans fats and saturated fatty acids.
Like saturated or animal fats, trans fats contribute to clogged arteries. Clogged arteries are a sign of heart disease; they increase your risk of both heart attack and stroke. Here's how it works: Trans fats raise low-density lipoprotein (LDL) or "bad" cholesterol levels. This contributes to the buildup of fatty plaque in arteries.
"The science that shows that trans fats increase LDL cholesterol levels is outstanding and very strong. All evidence is pointing in the same direction," Lichtenstein tells WebMD.
In the Nurse's Health Study, women who consumed the greatest amount of trans fats in their diet had a 50% higher risk of heart attack compared to women who consumed the least.
Some researchers suspect that trans fats also increase blood levels of two other artery-clogging compounds -- a fat-protein particle called lipoprotein(a) and blood fats called triglycerides.
Equally worrisome, population studies indicate that trans fats may raise the risk of diabetes. Researchers at the Harvard School of Public Health in Boston suggest that replacing trans fats in the diet with polyunsaturated fats (such as vegetable oils, salmon, etc.) can reduce diabetes risk by as much as 40%.
"Trans fats raise (bad) LDL cholesterol levels slightly less than do saturated fats," says Lichtenstein. "But saturated fats also raise levels of high density lipoprotein (HDL) or "good" cholesterol, and trans fatty acids don't." Trans fats may actually lower HDL.